2026 Vol. 8, No. 17
Human rabies vaccine has been in use for many years and has played a vital role in rabies prevention and control in China.
From 2021 to 2024, the overall reported rate of adverse events following immunization (AEFI) with human rabies vaccine was 18.57 per 100,000 doses, of which 96.29% were common vaccine reactions. Rare vaccine reactions occurred at a rate of 0.49 per 100,000 doses; allergic rash was the most frequently reported rare reaction and usually was non-serious. The rate of rare and serious vaccine reactions was 0.07 per 100,000 doses.
Human rabies vaccine, one of the most commonly used non-program vaccines in China, its AEFI rate fell within an acceptable range. Continued AEFI surveillance is essential for evaluating the post-marketing immnization safety of this vaccine.
Combination vaccines enhance health system efficiency and caregiver compliance by reducing the number of clinic visits and injections required for routine childhood immunization.
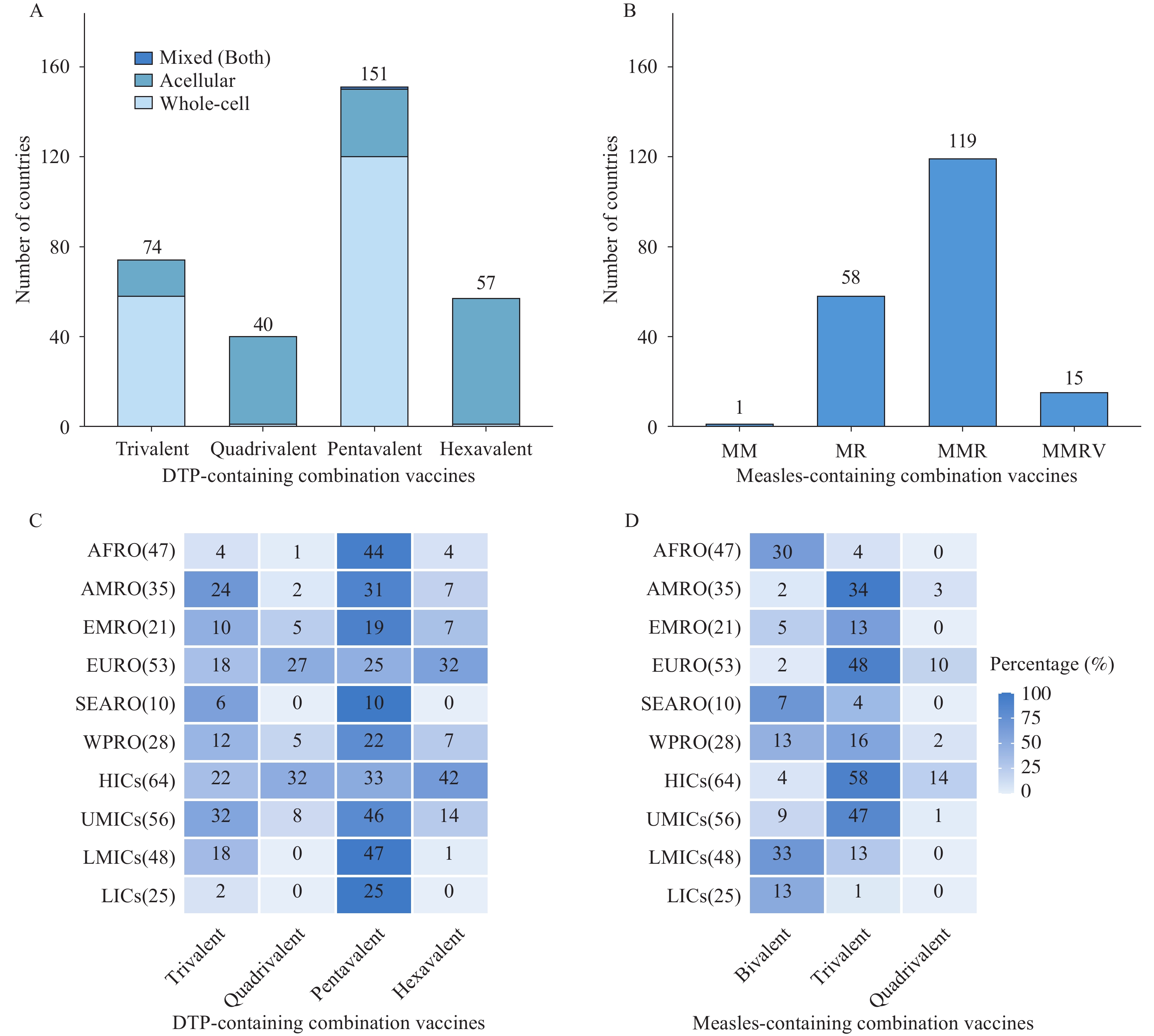
In 2024, eight types of combination vaccines were in use worldwide, grouped into two categories: diphtheria-tetanus-pertussis (DTP)-containing vaccines (trivalent, quadrivalent, pentavalent, and hexavalent) and measles-containing vaccines (two bivalent types, trivalent, and quadrivalent). Pentavalent DTP-containing vaccines and measles-mumps-rubella (MMR) vaccines were the most widely used globally, yet 15 countries still lacked any measles-containing combination vaccine. Significant coverage gaps and service drop-offs persisted in low-income countries in Africa and the Americas, reflecting economic disparities in access to advanced formulations such as hexavalent vaccines.
To align with Immunization Agenda 2030 (IA2030), tailored strategies should integrate combination vaccines with strengthened primary healthcare delivery and sustainable financing mechanisms. This approach is essential for improving service continuity and reducing dropout rates in underperforming regions.
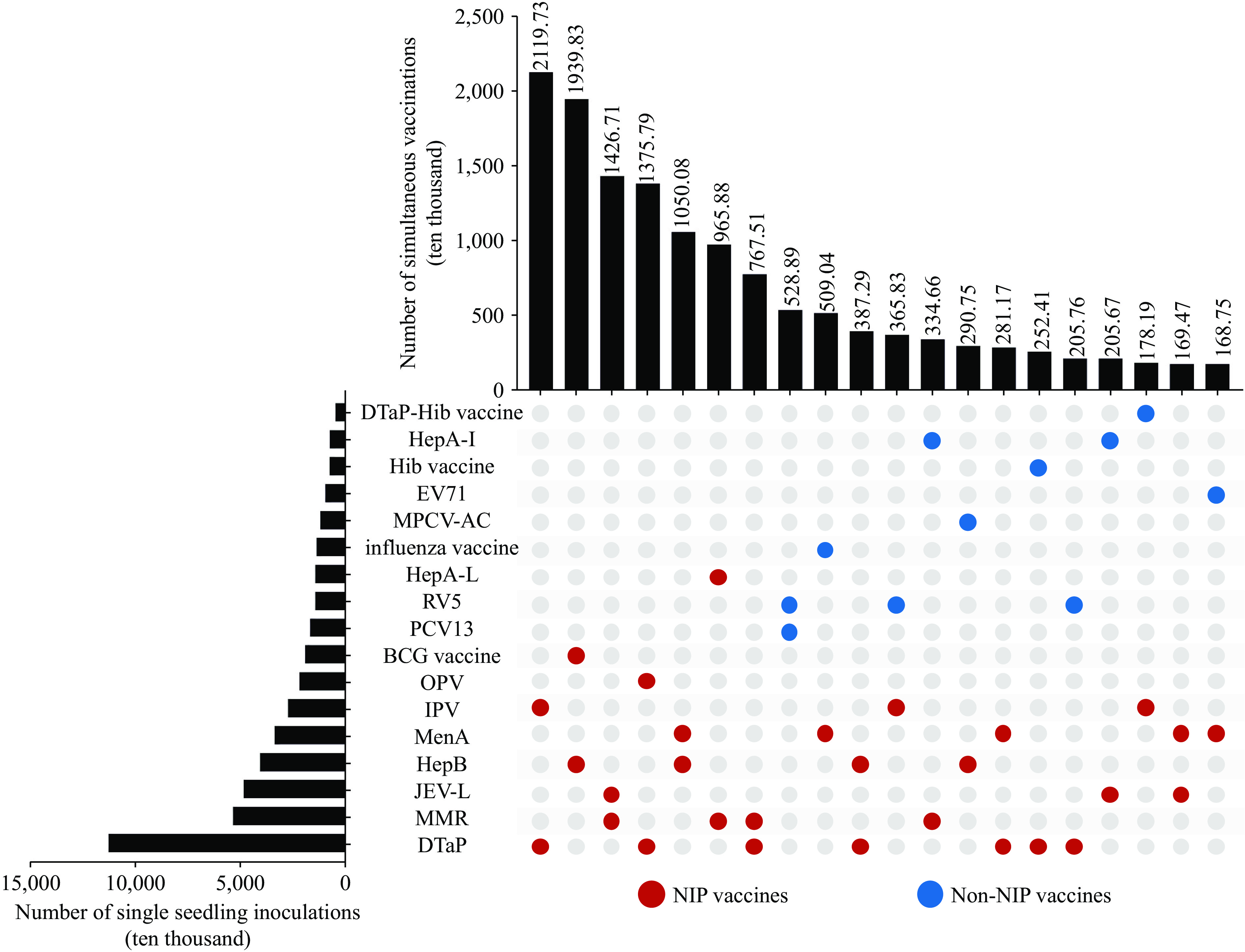
Simultaneous vaccination improves coverage and reduces clinic visits. In China, administering multiple vaccines during a single visit is widely practiced; studies have evaluated the safety and immunogenicity of specific vaccine combinations. However, systematic nationwide analyses remain limited.
During 2022–2024, 46.27% of vaccinations involved two vaccines administered simultaneously, 2.36% involved three, and 0.003% involved four or more. Most two-vaccine combinations included only National Immunization Program (NIP) vaccines, while combinations of three or more often included both NIP and non-NIP vaccines.
Real-world vaccination patterns provide evidence to inform research on multi-vaccine administration and combined vaccine development, supporting improvements in immunization service efficiency.
Anti-rabies monoclonal antibodies in combination with rabies vaccines have demonstrated safety and immunogenicity as alternatives to rabies immunoglobulins for category III post-exposure prophylaxis in adults.
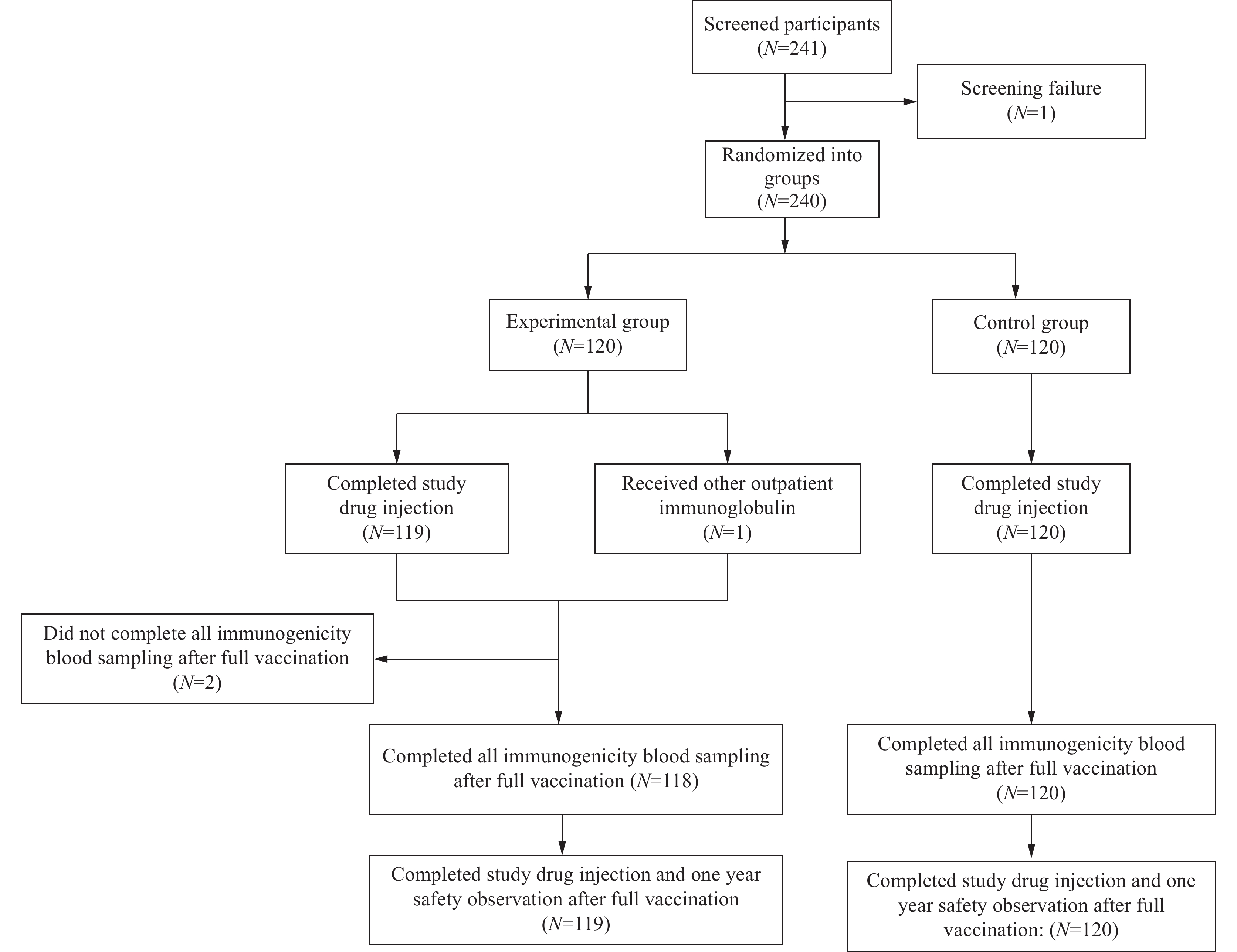
This randomized, double-blind, active-control phase III trial demonstrates the immunogenicity and safety of an anti-rabies monoclonal antibodies in individuals aged <18 years with category III rabies exposure.
These findings support the use of recombinant human monoclonal antibodies in pediatric populations and their potential role as alternative passive immunization options for category III rabies post-exposure prophylaxis in individuals aged <18 years.
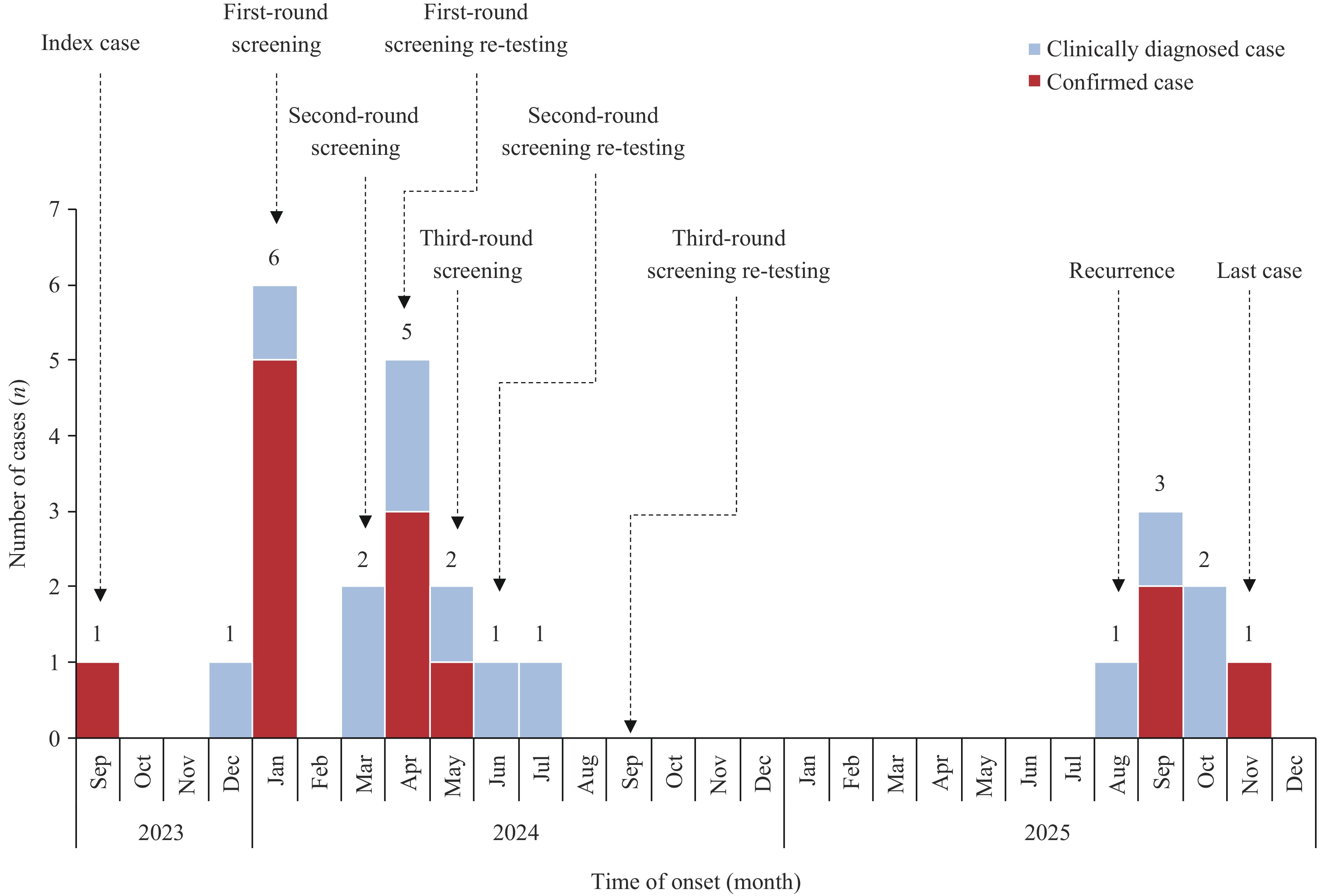
Tuberculosis (TB) is prone to cause clustered transmission in schools.
Following outbreak, the university conducted terminal disinfection of epidemic sites; however, Mycobacterium tuberculosis was detected (including air conditioner filters and air). Individuals with latent TB infection who receive preventive immunotherapy with Mycobacterium vaccae can develop active TB. Active cases were not identified promptly after preventive treatment, leading to renewed transmission.
Guidelines should add disinfection effectiveness evaluations and cover easily overlooked items, such as air conditioner filters. Follow-up examinations for individuals with LTBI who have completed preventive treatment remain necessary, as they still face a significant risk of developing active TB.



 Subscribe for E-mail Alerts
Subscribe for E-mail Alerts CCDC Weekly RSS Feed
CCDC Weekly RSS Feed