-
Introdution: To describe simultaneous vaccination among children aged 0–3 years in China during 2022–2024 and provide evidence to improve vaccination service efficiency and timely immunoprotection.
Methods: Individual-level vaccination records from the National Immunization Program Information System were analyzed for all vaccines administered to children aged 0–3 years nationwide from January 1, 2022, to December 31, 2024, focusing on same-visit vaccination practices.
Results: A total of 748.30 million vaccine doses were administered. Among these, 46.27% were given as two-vaccine simultaneous combinations, 2.36% as three-vaccine combinations, and 0.003% as four or more vaccines. Two-vaccine combinations mainly involved only National Immunization Program (NIP) vaccines (74%–78%), whereas three- and four-vaccine combinations more often included both NIP and non-NIP vaccines. Common combinations included diphtheria, tetanus, and acellular pertussis (DTaP) with inactivated poliovirus vaccine (IPV), HepB, or BCG.
Conclusion: Simultaneous vaccination in China is predominantly characterized by the co-administration of two vaccines, with three or more used less frequently. These findings provide evidence to guide optimization of immunization practices, research prioritization, and improvements in vaccination efficiency.
-
Vaccination is among the most cost-effective strategies for preventing infectious diseases (1). Children aged 0-3 years are a priority population in China’s immunization programs, and timely, safe vaccination is essential for maximizing protection and program impact. As the number and variety of recommended vaccines increase, simultaneous vaccination, the administration of two or more different vaccines during the same visit, has become an important approach for optimizing immunization schedules (2). Widely used internationally when safety and immunogenicity requirements are met, this practice improves coverage and reduces clinic visits (3). In China, simultaneous vaccination is recommended: vaccines included in the National Immunization Program (NIP vaccines) may be administered simultaneously according to routine and catch-up schedules. Parents may also choose non-NIP vaccines in place of NIP vaccines, expanding the combinations eligible for simultaneous administration (4–5). Although studies in China have evaluated the safety and immunogenicity of specific combinations (6–8), nationwide real-world descriptions remain limited. This study uses National Immunization Program Information System (NIPIS) data to describe same-visit administration of two, three, and four or more vaccines among children aged 0–3 years in China during 2022–2024.
Individual-level vaccination records for children aged 0–3 years vaccinated between January 2022 and December 2024 across 32 provincial-level administrative divisions (PLADs) in China were extracted from NIPIS. Simultaneous vaccination was defined as administration of two or more different vaccines during one clinic visit at separate anatomical sites (9). Combinations were classified as NIP-only, non-NIP-only, or mixed NIP and non-NIP vaccines.
NIP vaccines included hepatitis B (HepB), Bacillus Calmette-Guérin (BCG), inactivated poliovirus vaccine (IPV), oral poliomyelitis attenuated live vaccine (OPV), DTaP, measles-mumps-rubella (MMR), Japanese encephalitis attenuated live vaccine (JEV-L), group A meningococcal polysaccharide vaccine (MenA), group A-C meningococcal polysaccharide vaccine (MenAC), inactivated hepatitis A (HepA-I), and hepatitis A live attenuated vaccine (HepA-L). Non-NIP vaccines included trivalent rotavirus vaccine, pentavalent rotavirus vaccine, rotavirus vaccine, 13-valent pneumococcal conjugate vaccine (PCV13), 23-valent pneumococcal polysaccharide vaccine (PPSV23), DTaP-Hib vaccine, DTaP-IPV/Hib vaccine, Enterovirus Type 71 Vaccine (EV71), group A and C meningococcal polysaccharide conjugate vaccine (MPCV-AC), ACYW135 meningococcal polysaccharide vaccine, ACYW135 meningococcal conjugate vaccine, and Influenza vaccine, varicella vaccine, Hib vaccine, Japanese Encephalitis Inactivated Vaccine (JEV-I), Measles and Mumps vaccine(MM), etc. Quadrivalent and pentavalent vaccines were considered as single vaccines. COVID-19 vaccines were excluded because they were not licensed for children aged 0–3 years during the study period.
Descriptive statistics summarized simultaneous vaccination practices, with categorical variables presented as percentages. Data were curated using Microsoft Excel (version 2016, Microsoft Office, Washington, USA) and analyzed with R (version 4.2.1, R Statistical Computing Foundation, Vienna, Austria).
During 2022–2024, 748.30 million vaccine doses were administered to children aged 0–3 years. Among these, 346.21 million (46.27%) involved simultaneous administration of two vaccines, 17.68 million (2.36%) involved three vaccines, 24,460 (0.003%) involved four or more vaccines (Table 1).
Year Single vaccine Two vaccines Three vaccines ≥4 vaccines Total doses
N (million)
Proportion (%)N (million) Proportion (%) N (million) Proportion (%) N (million) Proportion (%) N Proportion (%) 2022 130.94 50.47 121.66 46.89 6.84 2.64 9,840 0.004 259.46 100 2023 129.99 50.13 122.62 47.29 6.68 2.58 9,100 0.004 259.30 100 2024 123.45 53.78 101.93 44.41 4.16 1.81 5,520 0.003 229.54 100 Total 384.39 51.36 346.21 46.27 17.68 2.36 24,460 0.003 748.30 100 Table 1. Simultaneous administration of two or more vaccines during a visit to children aged 0–3 years in China, 2022–2024.
Overall, 363.91 million doses were administered simultaneously. Two-vaccine combinations accounted for 95.14% of these doses, with 75.02% involving pairs of NIP vaccines. Three-vaccine combinations represented 4.86%, of which 53.82% included both NIP and non-NIP vaccines. Administration of four or more vaccines occurred in less than 0.01% of all doses (Table 2).
Year Two vaccines Three vaccines ≥4 vaccines Only NIP vaccines N (million doses) (%) Only non-NIP vaccines N (million doses) (%) Mixed combination N (million doses) (%) Only NIP vaccines N (million doses) (%) Only Non-NIP vaccines N (million doses) (%) Mixed combination N (million doses) (%) Only NIP vaccines N (doses) (%) Only non-NIP vaccines N (doses) (%) Mixed combination N (doses) (%) 2022 90.12 (74.07) 8.41 (6.91) 23.13 (19.01) 2.87 (42.00) 0.23 (3.37) 3.74 (54.62) 1,700 (17.60) 540 (5.50) 7,600 (76.90) 2023 90.70(73.97) 8.71 (7.10) 23.21 (18.93) 2.68 (40.12) 0.29 (4.41) 3.70 (55.47) 1,900 (21.08) 400 (4.37) 6,800 (74.55) 2024 78.90 (77.41) 7.06 (6.93) 15.96 (15.66) 1.92 (46.24) 0.16 (3.90) 2.07 (49.86) 1,700 (31.34) 20 (0.36) 3,800 (68.30) Total 259.72 (75.02) 24.18 (6.98) 62.30 (17.99) 7.48 (42.29) 0.69 (3.89) 9.52 (53.82) 5,300 (21.67) 960 (3.92) 18,200 (74.41) Abbreviation: NIP=national immunization program. Table 2. Vaccines administered together by study year, combination type, and number of vaccines administered at a visit.
The seven most common two-vaccine combinations were pairs of NIP vaccines (Figure 1). The most frequent pairings were DTaP with IPV at three and four months of age, BCG with HepB at birth, and MMR with JEV-L at 8 months (Figure 1). The most common mixed combinations were MenA with influenza vaccine and RV5 with IPV.
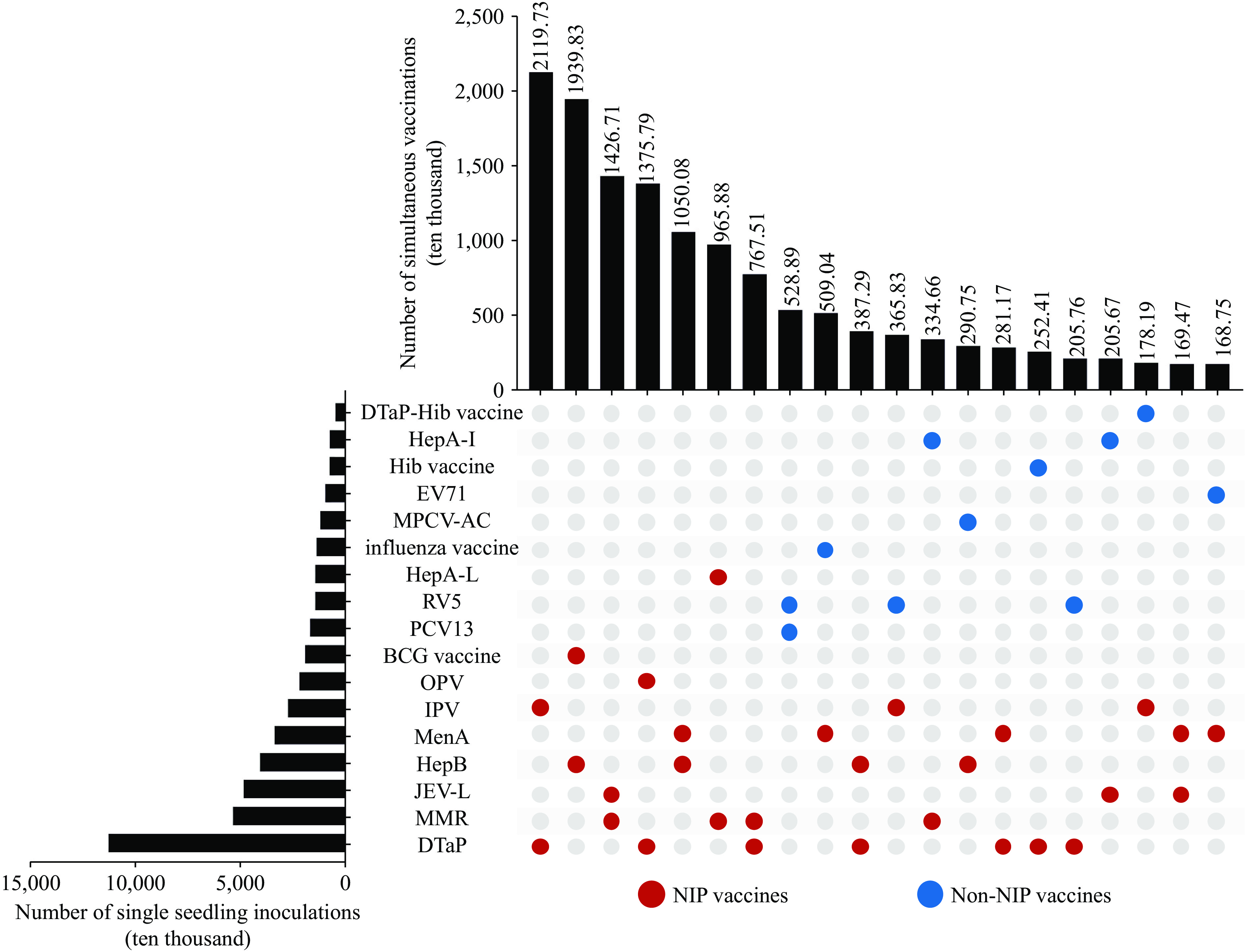 Figure 1.
Figure 1.Vaccine combinations for simultaneous administration of two vaccines.
Abbreviation: DTaP-Hib=diphtheria, tetanus, acellular pertussis and Haemophilus influenzae type b combined vaccine; HepA-I=hepatitis A inactivated vaccine; Hib vaccine=Haemophilus influenzae type b vaccine; EV71=Enterovirus Type 71 Vaccine, Inactivated; MPCV-AC=group A and group C meningococcal polysaccharide conjugate vaccine; HepA-L=hepatitis A live attenuated vaccine; RV5=pentavalent rotavirus vaccine; PCV13=pneumococcal polysaccharide conjugate vaccine (13-valent); BCG vaccine=Bacillus Calmette-Guérin vaccine; OPV=oral poliovirus vaccine; IPV=inactivated poliovirus vaccine; MenA=group A meningococcal vaccine; HepB=hepatitis B vaccine; JEV-L=Japanese encephalitis live attenuated vaccine; MMR=measles, mumps and rubella combined vaccine; DTaP=diphtheria, tetanus, and acellular pertussis vaccine.Among three-vaccine combinations, the most frequent were IPV, HepB, and BCG; IPV, DTaP, and pentavalent rotavirus vaccine; and OPV, HepB, and DTaP (Figure 2). For four-vaccine combinations, the most common were OPV, HepB, DTaP, and MenA; IPV, HepB, DTaP, MenA; and OPV, HepB, DTaP, and pentavalent rotavirus vaccine (Figure 3).
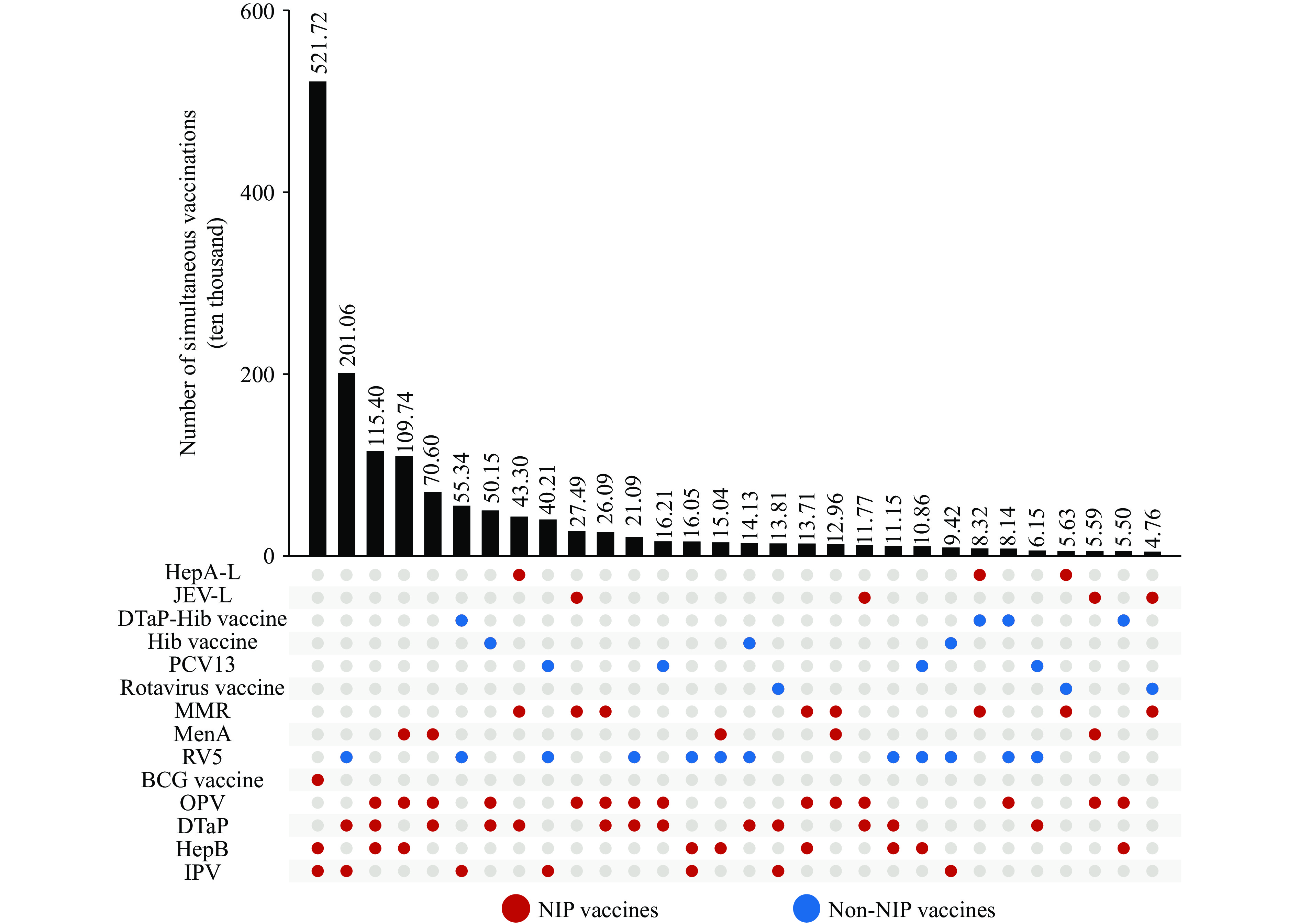 Figure 2.
Figure 2.Vaccine combinations for simultaneous administration of three vaccines.
Abbreviation: HepA-L=hepatitis A vaccine (live attenuated); JEV-L=Japanese encephalitis vaccine (live attenuated); DTaP-Hib vaccine=diphtheria, tetanus, acellular pertussis and Haemophilus influenzae type b combined vaccine; Hib vaccine=Haemophilus influenzae type b vaccine; PCV13=pneumococcal polysaccharide conjugate vaccine (13-valent); MMR=measles, mumps and rubella combined vaccine; MenA=group A meningococcal vaccine; RV5=pentavalent rotavirus vaccine;BCG vaccine=Bacillus Calmette-Guérin vaccine; OPV=oral poliovirus vaccine; DTaP=diphtheria, tetanus and acellular pertussis vaccine; HepB=hepatitis B vaccine; IPV=inactivated poliovirus vaccine.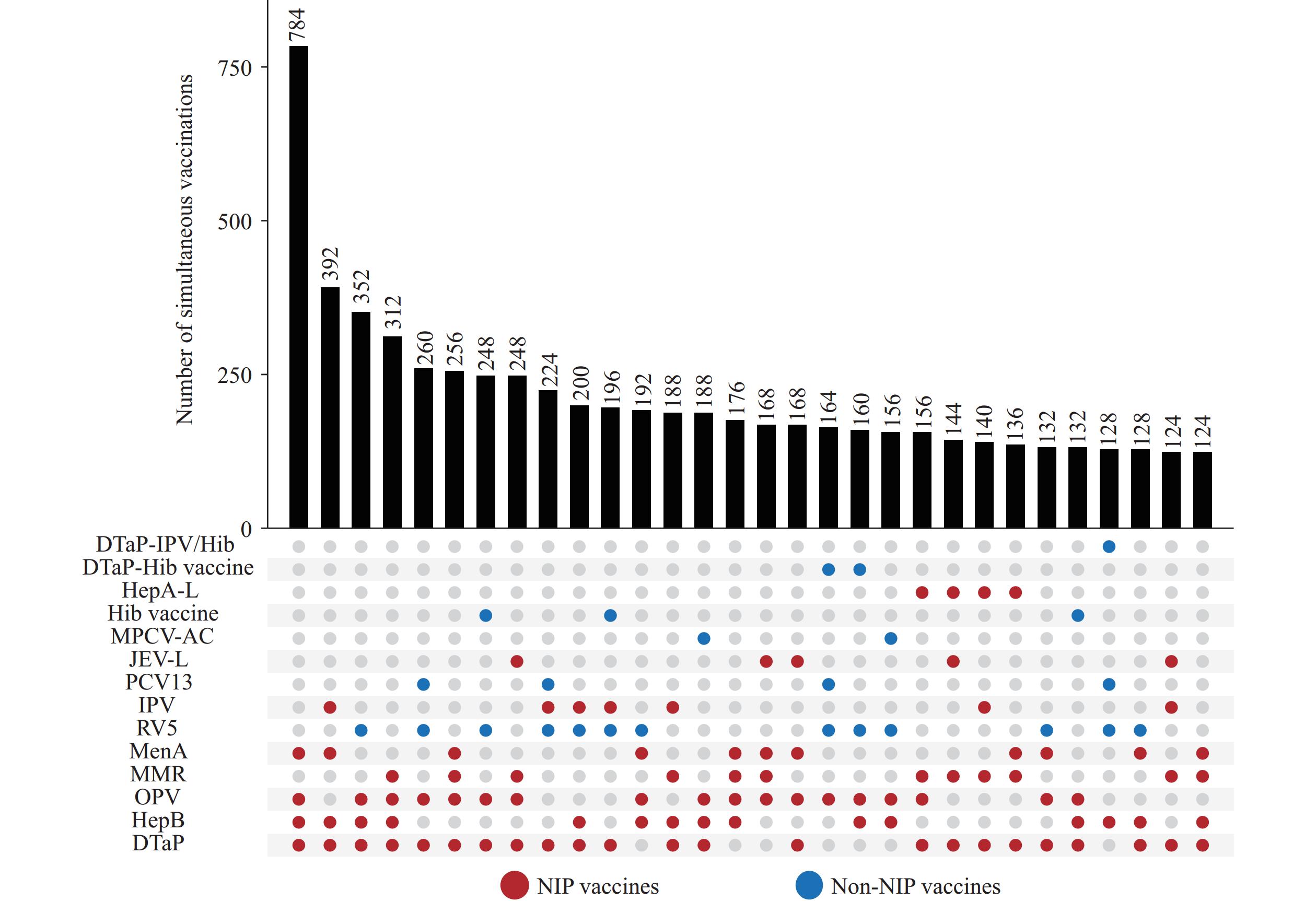 Figure 3.
Figure 3.Vaccine combinations for simultaneous administration of four or more vaccines.
Abbreviation: DTaP-IPV/Hib=diphtheria, tetanus, and acellular pertussis, inactivated poliovirus vaccine and Haemophilus influenzae type b combined vaccine; DTaP-Hib vaccine=diphtheria, tetanus, acellular pertussis and Haemophilus influenzae type b combined vaccine; HepA-L=hepatitis A live attenuated vaccine; Hib vaccine=Haemophilus influenzae type b vaccine; MPCV-AC=group A and group C meningococcal polysaccharide conjugate vaccine; JEV-L=Japanese encephalitis live attenuated vaccine; PCV13=pneumococcal polysaccharide conjugate vaccine (13-valent); IPV=inactivated poliovirus vaccine; RV5=pentavalent rotavirus vaccine; MenA=group A meningococcal vaccine; MMR=measles, mumps and rubella combined vaccine; OPV=oral poliovirus vaccine; HepB=hepatitis B vaccine; DTaP=diphtheria, tetanus and acellular pertussis vaccine. -
This study used vaccination records of children aged 0–3 years during 2022–2024 in the NIPIS to describe real-world simultaneous vaccination practices in China. Simultaneous vaccination was common, with two vaccines administered during the same visit as the predominant pattern, whereas three or more vaccines were administered much less frequently. Some simultaneous vaccination events involved both NIP and non-NIP vaccines, indicating concurrent use of different vaccine categories in routine practice.
The predominance of two-vaccine administration likely reflects clustering within China’s national immunization schedule. Several vaccines are scheduled at the same or adjacent ages, at birth and during early infancy (e.g., at 2 months, 3–4 months, and 6–8 months of age), creating opportunities for simultaneous administration. Common combinations included BCG with hepatitis B at birth and DTaP with IPV during infancy. These pairings align with the national schedule and may support adherence, reduce missed doses, and lower service burdens for families and clinics.
In contrast, simultaneous administration of three or more vaccines was far less frequent. Increasing the number of vaccines administered during a single visit raises operational demands and requires vaccinators to manage contraindications and precautions across multiple vaccines (10). Parental acceptance also influences implementation. Previous studies suggest parents generally accept two simultaneous vaccines, whereas willingness declines when three or more are administered, often due to concerns about discomfort, adverse reactions, or effectiveness (11–13).
Despite the low overall proportion, three-vaccine combinations showed recognizable patterns. Most involved NIP vaccines combined with a single non-NIP vaccine, such as DTaP, IPV, and rotavirus vaccine, typically at ages when vaccination schedules overlapped. This pattern suggests that some three-vaccine strategies are adopted when schedule requirements and children’s vaccination needs coincide.
As the number of vaccines increased, combinations consisting only of non-NIP vaccines were uncommon, whereas those including NIP vaccines were more frequent. This pattern may reflect the self-paid nature and accessibility of non-NIP vaccines, as well as financial considerations. In addition, technical guidance on simultaneous administration of some non-NIP vaccines remains limited (2), which may encourage cautious implementation by clinics. Consequently, non-NIP vaccines are often administered alongside NIP vaccines rather than concentrated in a single visit.
Simultaneous administration of four or more vaccines was extremely rare. This finding may indicate a practical upper limit on the number of vaccines administered during one visit under current service conditions and levels of parental acceptance. Future adjustments to immunization schedules or the introduction of new vaccines may therefore require balancing increased clinic visits with greater simultaneous administration unless combination vaccines reduce injection numbers.
An important contribution of this study lies in its use of nationwide vaccination records to disaggregate simultaneous vaccination events by number and combination type, identifying recurring patterns in routine practice. Such structural analysis of actual vaccination behavior may help define priorities for studies on the safety, reactogenicity, and immunogenicity of multi-vaccine administration. The stable combination patterns observed may also inform vaccine product optimization and the development of combination vaccines to improve immunization efficiency by reducing injections.
Several limitations should be noted. First, as a retrospective observational study, it could not assess parental or vaccinator knowledge, attitudes, or preferences in vaccination decision-making. Second, reliance on immunization information system records may introduce incomplete entries or data entry errors.
In summary, simultaneous vaccination among young children aged 0-3 years in China is characterized by two-vaccine administration as the dominant pattern, with three or more vaccines administered under specific conditions. By describing vaccine numbers and combination structures using real-world data, this study provides a foundation for optimizing immunization practices, guiding targeted research, and improving vaccination efficiency.
-
All staff members who contributed to data collection and participated in this study.
-
The Survey on the Current Status of Simultaneous Vaccination received approval from the Chinese Center for Disease Control and Prevention Institutional Review Board in May 2025 (Approval Notice No. 202515).
HTML
| Citation: |



 Download:
Download:




