-
Introduction: Optimizing and streamlining immunization schedules through the use of combination vaccines can advance post-pandemic vaccination recovery and “The Big Catch-up” initiative. This study assessed the global landscape of combination vaccine use and its association with immunization coverage in 2024.
Methods: Using 2024 World Health Organization/United Nations International Children's Emergency Fund (WHO/UNICEF) Joint Reporting Form (JRF) data and official national coverage estimates, we conducted a cross-sectional descriptive analysis of diphtheria-tetanus-pertussis (DTP)-containing and measles-containing combination vaccine use patterns; countries were stratified by WHO region and World Bank income classification.
Results: In 2024, eight types of combination vaccines were in use worldwide, divided into two categories: DTP-containing vaccines, including trivalent, quadrivalent, pentavalent, and hexavalent formulations; and measles-containing vaccines, including two bivalent types, trivalent, and quadrivalent formulations. Among DTP-containing vaccines, pentavalent formulations were the most widely used globally (151 of 194 Member States), while hexavalent vaccine use was concentrated in high-income countries. The trivalent formulation was the most commonly used measles-containing combination vaccine (119 countries), but 15 countries relied exclusively on monovalent measles vaccine. Significant regional and economic disparities were observed: low-income countries in Africa and the Americas experienced substantial coverage drop-off between the first and third DTP doses, suggesting service continuity bottlenecks despite high initial access.
Conclusion: Although combination vaccines are widely used, their potential to bridge immunization equity gaps remains unrealized due to systemic barriers in low-resource settings. Strengthening primary healthcare and ensuring sustainable financing for advanced vaccine formulations are essential to achieving global immunization targets.
-
Routine immunization ranks among the most cost-effective public health interventions for preventing and controlling vaccine-preventable diseases (VPDs). However, the coronavirus disease 2019 (COVID-19) pandemic caused unprecedented disruptions to routine immunization services worldwide (1–2). In the context of post-pandemic immunization recovery and implementation of The Big Catch-up initiative, optimizing immunization schedules and promoting combination vaccine use are widely recognized as key strategies to strengthen health system resilience and improve vaccination service performance (3). By conferring protection against multiple diseases in a single visit, combination vaccines reduce the vaccination burden on caregivers while enabling more efficient allocation of limited health resources through fewer required clinic visits — particularly in settings with constrained service capacity (4).
Despite the well-established clinical advantages of combination vaccines, previous studies have largely focused on single-country analyses or evaluations of specific products (5–6). Few systematic assessments have examined global patterns of combination vaccine use and their association with immunization coverage recovery. Moreover, inherent reporting lags in global vaccine utilization databases mean that analyses reflecting the most recent data (2024) have not yet been reported. Using 2024 global monitoring data, this study aims to systematically assess the status of combination vaccine use worldwide and explore its potential associations with immunization coverage recovery, thereby providing evidence to inform the development of more resilient immunization strategies in the post-pandemic era.
This cross-sectional descriptive study analyzed publicly available, country-level data to systematically assess combination vaccine use patterns in routine childhood immunization programs across 194 WHO Member States and to examine the association between these patterns and immunization coverage.
Vaccine use data were extracted from the 2024 WHO/UNICEF Joint Reporting Form (JRF) (7), which documents vaccine types, formulations, and schedules implemented in national routine childhood immunization programs. Coverage data were obtained from the 2024 official national coverage estimates published by UNICEF (8). The two datasets were merged by country code. Countries and areas that reported complete routine childhood immunization information in the 2024 JRF and had corresponding coverage estimates were included in the final analysis.
Based on antigen components reported in the JRF, combination vaccines were categorized into two main groups: DTP-containing and measles-containing combination vaccines. DTP-containing combination vaccines were defined as those used in routine immunization that include diphtheria, tetanus, and pertussis antigens, either alone or combined with additional antigens such as hepatitis B, Haemophilus influenzae type b (Hib), or inactivated poliovirus vaccine (IPV). Measles-containing combination vaccines were defined as those containing measles vaccine virus in combination with at least one other vaccine virus — rubella, mumps, or varicella.
Three indicators were used to assess immunization program performance: coverage with the first dose of DTP-containing vaccine (DTP1), as an indicator of access to basic immunization services; coverage with the third dose of DTP-containing vaccine (DTP3), reflecting completion of the primary series and service continuity; and coverage with the first dose of measles-containing vaccine (MCV1), serving as a core indicator of control capacity for highly transmissible diseases and overall program reach.
To examine global disparities, countries were stratified by WHO region (geographic dimension) (9) and World Bank income classification for the 2026 fiscal year based on 2024 financial data (economic dimension) (10). All analyses were descriptive, intended to characterize current patterns and distributions without causal inference.
Four types of DTP-containing combination vaccines were in use: trivalent (DTwP or DTaP), quadrivalent (DTaP-IPV), pentavalent (DTwP-Hib-HepB or DTaP-Hib-IPV), and hexavalent (DTaP-Hib-HepB-IPV). Among these, pentavalent vaccines were the most widely used formulations (Figure 1). By 2024, 151 countries had introduced pentavalent vaccines into routine immunization programs, with 120 using whole-cell pertussis (wP) formulations, 30 using acellular pertussis (aP) formulations, and 1 country using both. Quadrivalent and hexavalent vaccine use was largely concentrated in countries employing acellular pertussis formulations, indicating a technological link between formulation type and valency.
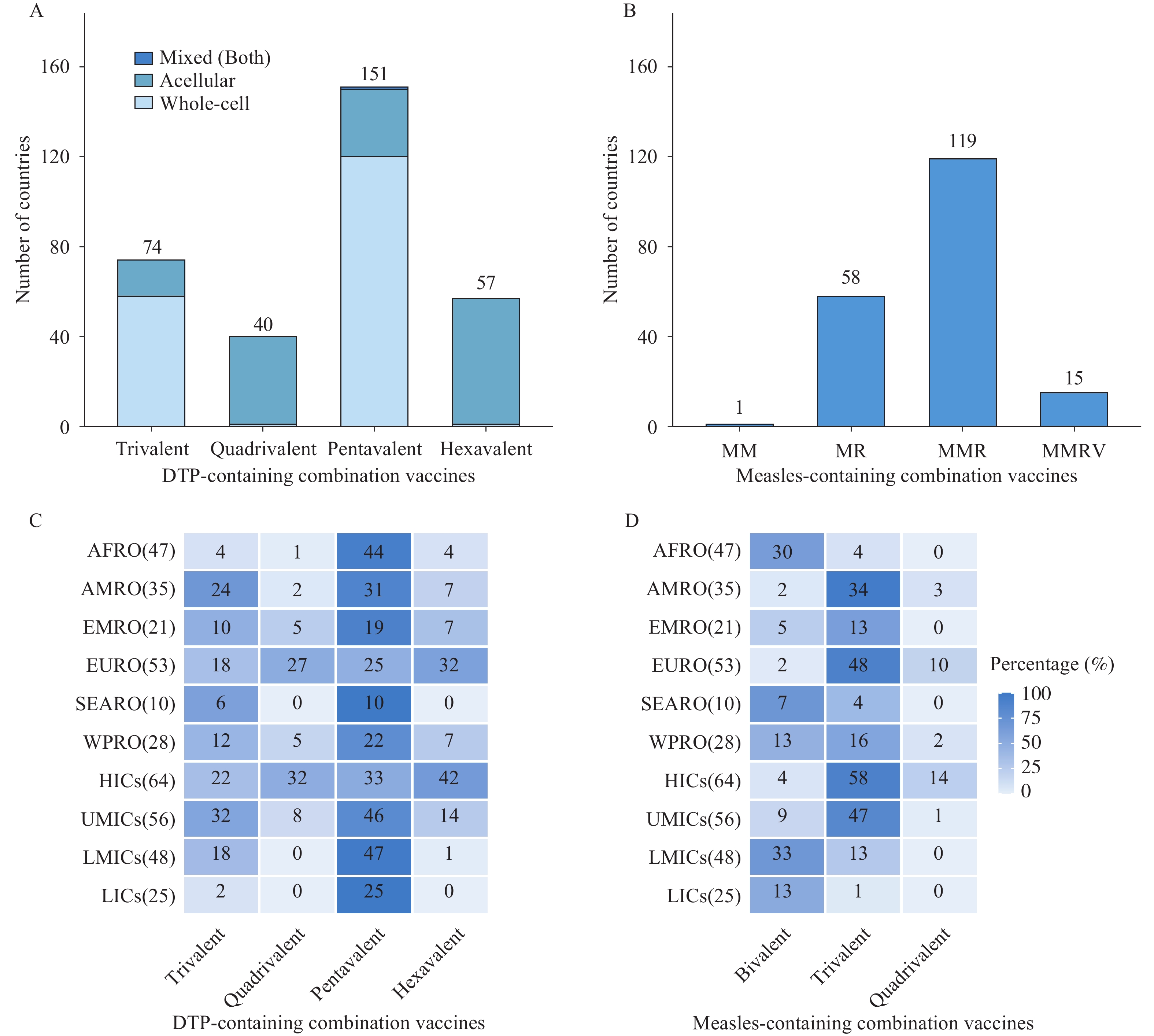 Figure 1.
Figure 1.Number of countries using different combination vaccines in routine childhood immunization, 2024. (A) Number of countries using various DTP-containing vaccines; (B) Number of countries using various measles-containing vaccines; (C) Distribution of DTP-containing vaccines by WHO region and income level; (D) Distribution of measles-containing vaccines by WHO region and income level.
Abbreviation: DTP=diphtheria-tetanus-pertussis; MM=measles-mumps; MR=measles-rubella; MMR=measles-mumps-rubella; MMRV=measles-mumps-rubella-varicella; AFRO=Regional Office for Africa; AMRO=Regional Office for the Americas; EMRO=Regional Office for the Eastern Mediterranean; EURO=Regional Office for Europe; SEARO=Regional Office for South-East Asia; WPRO=Regional Office for the Western Pacific; HICs=High-income countries; UMICs=Upper-middle-income countries; LMICs=Lower-middle-income countries; LICs=Low-income countries.Four types of measles-containing combination vaccines were in use: measles-mumps (MM), measles-rubella (MR), measles-mumps-rubella (MMR), and measles-mumps-rubella-varicella (MMRV). For measles prevention and control, MMR was the predominant global choice, incorporated into routine immunization programs in 119 countries. Fifty-eight countries used MR and 15 countries used MMRV. Fifteen countries exclusively used monovalent measles vaccines and, as of 2024, had not yet introduced any measles-containing combination vaccine — all were low- and middle-income countries, primarily in Africa and the Middle East. Continued reliance on monovalent measles vaccine in these settings highlights an opportunity to strengthen rubella and mumps control through adoption of combination formulations.
Significant heterogeneity emerged in combination vaccine use patterns across WHO regions and income groups (Figure 1). In the African Region (AFRO) and the Eastern Mediterranean Region (EMRO), pentavalent vaccines predominated among DTP-containing formulations, with limited adoption of higher-valent options such as hexavalent vaccines. Countries in the European Region (EURO) and the Western Pacific Region (WPRO) more frequently used higher-valent DTP-containing combination vaccines. The Region of the Americas (AMRO) and the South-East Asia Region (SEARO) appeared to be in transitional stages, with gradual expansion toward higher-valent combinations.
Use of measles-containing combination vaccines correlated positively with national income level. High-income countries (HICs) and upper-middle-income countries (UMICs), particularly in the EURO and WPRO regions, widely implemented trivalent or quadrivalent formulations. In contrast, low-income countries (LICs) and lower-middle-income countries (LMICs) continued to rely on bivalent or monovalent measles vaccines. MMRV vaccine use was greatest in HICs and nearly absent in LICs, underscoring the critical role of economic capacity in determining access to technologically advanced vaccines.
Global immunization coverage data for 2024 revealed substantial regional and economic disparities (Figure 2). Worldwide, DTP1 coverage was generally high, indicating broad initial access to immunization services. However, pronounced regional variation in DTP3 and MCV1 coverage pointed to marked differences in the completion of full vaccination schedules.
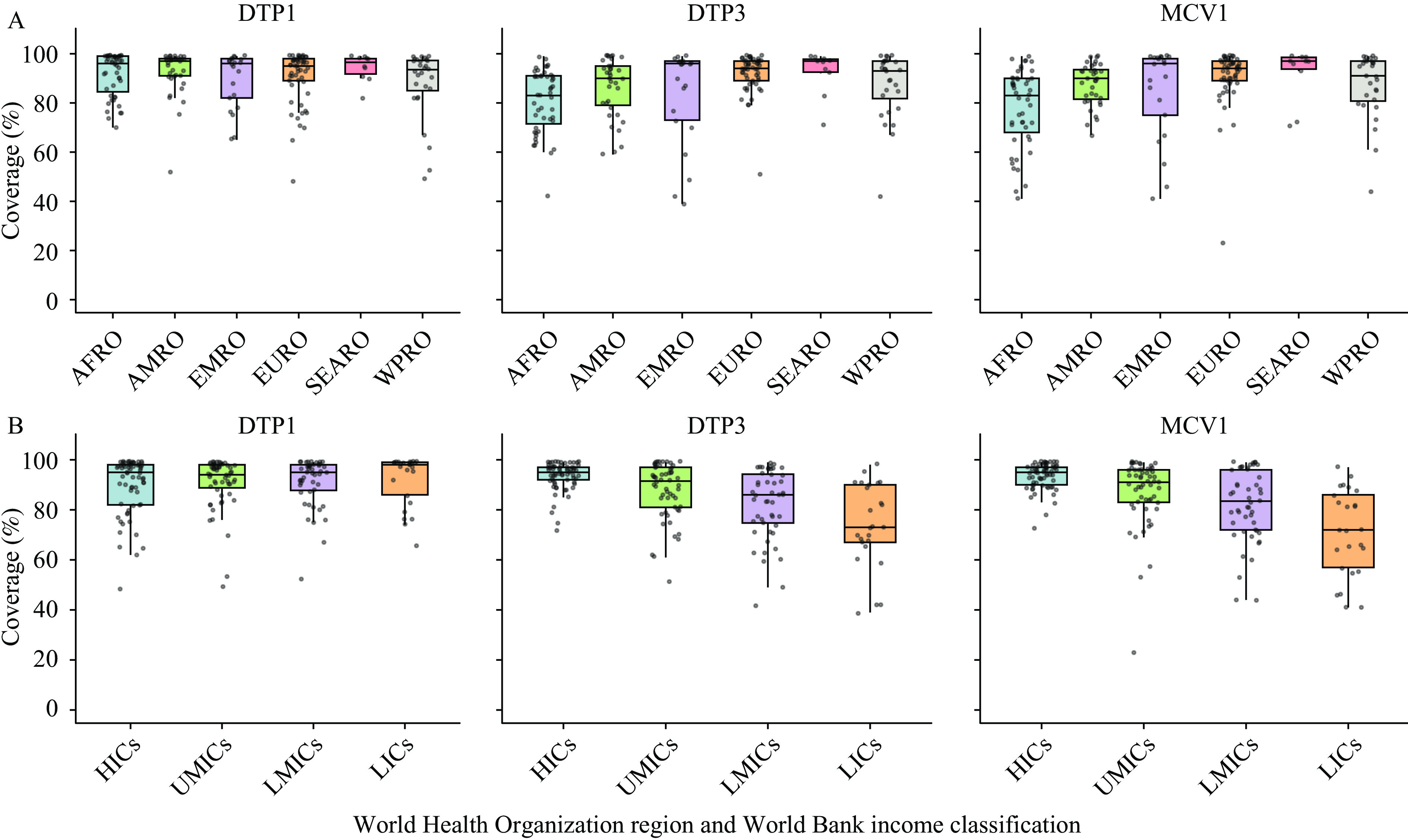 Figure 2.
Figure 2.Vaccination coverage of DTP1, DTP3, and MCV1 by WHO region and income level in 2024. (A) Distribution of DTP1, DTP3, and MCV1 coverage across WHO regions; (B) Distribution of DTP1, DTP3, and MCV1 coverage across income levels.
Abbreviation: DTP1=diphtheria-tetanus-pertussis, first dose; DTP3=diphtheria-tetanus-pertussis, third dose; MCV1=measles-containing vaccine, first dose; AFRO=Regional Office for Africa; AMRO=Regional Office for the Americas; EMRO=Regional Office for the Eastern Mediterranean; EURO=Regional Office for Europe; SEARO=Regional Office for South-East Asia; WPRO=Regional Office for the Western Pacific; HICs=High-income countries; UMICs=Upper-middle-income countries; LMICs=Lower-middle-income countries; LICs=Low-income countries.The EMRO, EURO, SEARO, and WPRO regions maintained high DTP3 and MCV1 coverage levels, with low dropout rates between DTP1 and DTP3 and relatively small inter-country variation. In contrast, the AFRO and AMRO regions experienced substantial declines from DTP1 to DTP3 coverage alongside lower overall MCV1 coverage, accompanied by marked inter-country disparities. HICs and UMICs consistently achieved high, stable coverage across all three indicators, whereas LMICs and LICs exhibited lower overall coverage and wider gaps between DTP1 and DTP3, suggesting bottlenecks in service continuity. DTP3 and MCV1 coverage displayed clear stepwise gradients across income groups, with greater dispersion and inequality in non-high-income groups.
-
This study provides a comprehensive overview of global combination vaccine use in 2024, highlighting significant disparities across WHO regions and income groups. While DTP1 coverage remains relatively robust worldwide, the pronounced drop-off between DTP1 and DTP3 — particularly in AFRO and AMRO — underscores persistent challenges in health system retention and service continuity. These findings align with the “backsliding” trends observed in the post-pandemic era, during which immunization coverage recovery has been uneven (11−12). Widespread adoption of combination vaccines, including pentavalent and hexavalent formulations, represents a critically important strategy to mitigate these gaps by reducing the number of injections and potential dropout points, consistent with the Immunization Agenda 2030 (IA2030) priority of leaving no one behind (13).
Our analyses demonstrate that HICs and UMICs maintained high, stable DTP3 and MCV1 coverage rates, largely benefiting from mature immunization programs and extensive hexavalent vaccine use. In contrast, LICs and LMICs experienced significant coverage gaps and higher dropout rates. This economic gradient suggests that, despite support from global initiatives such as Gavi, the Vaccine Alliance, financial and logistical barriers remain significant obstacles. Recent studies have shown that introducing combination vaccines in LICs effectively improves timely vaccination rates, yet supply chain fragility and workforce shortages continue to prevent realization of their full potential (14). Furthermore, the disparities highlight the particular vulnerability of middle-income countries that have transitioned out of Gavi or other donor support (15). Notably, regional variation in priority diseases included in immunization programs may drive differences in combination vaccine adoption, and disparities in economic capacity further influence the range of diseases that public funding can cover.
Maximizing combination vaccine use extends beyond product introduction — it demands strengthening underlying health systems. The substantial gap between DTP1 and DTP3 in specific regions indicates that, while initial access is achievable, retaining children in the system to complete the immunization schedule remains a key bottleneck. Combination vaccines offer a major advantage: by delivering multiple antigens in a single visit, they maximize the protective impact of every contact between the child and the health system. This is particularly relevant for “zero-dose” and under-immunized children in conflict-affected or remote settings, as emphasized by “The Big Catch-up” initiative launched in 2023 (16). The value of combination vaccines should therefore be re-evaluated at the systemic level, recognizing them as essential tools for enhancing immunization service efficiency, improving population compliance, and optimizing overall immunization outcomes.
Although combination vaccines offer significant advantages, their widespread adoption in low-income countries remains constrained by multiple barriers, including economic, supply chain, and infrastructure challenges. Financial limitations and stringent cold chain requirements prevent resource-limited regions from absorbing the high upfront costs. Context-specific strategies are therefore needed, including alleviating financial pressures through international tiered pricing and joint procurement, promoting technology transfer for regional production, and prioritizing introduction in areas with high dropout rates to maximize health benefits per dose.
The global disparities revealed by this study offer important insights for China’s immunization program development. As a country transitioning toward a higher-level public health system, China urgently needs to improve immunization service efficiency and optimize vaccination delivery models. Experience from high-income countries demonstrates that advanced combination vaccines enhance system efficiency and sustain high coverage levels. Against this backdrop, China should strengthen its combination vaccine development and production capabilities while encouraging domestic enterprises to increase investment, thereby reducing reliance on external supply chains and enhancing vaccine accessibility and affordability to better serve the National Immunization Program (NIP). Simultaneously, aligning vaccine innovation with the Expanded Program on Immunization (EPI) will drive the transformation of immunization services toward a more efficient, integrated model. By bolstering combination vaccine development and production systems, China can meet domestic public health needs while playing a more active role in the global vaccine supply system, thereby advancing immunization equity.
Findings in this report are subject to at least four limitations. First, the analysis relied on aggregated national-level data, which may obscure subnational inequalities in immunization coverage and mask localized pockets of under-immunization. Second, as 2024 represents a critical stage in the post-pandemic recovery of routine immunization services, the long-term stability of the observed patterns requires continued monitoring. Third, since only 2024 data were analyzed, tracking temporal changes in global combination vaccine use was not possible. Finally, this study describes current patterns and distributions of combination vaccine use and immunization coverage; its cross-sectional design limits causal inference.
In conclusion, while combination vaccines were widely used in 2024, their potential to close the immunization equity gap has not been fully realized due to systemic challenges. To align with IA2030 and global vaccination targets, tailored strategies are needed. For lower-performing regions, integrating combination vaccines with strengthened primary healthcare delivery and community engagement is essential. Policymakers should prioritize sustainable financing and supply chain optimization to ensure that the efficiency advantages of combination vaccines translate into tangible improvements in full, timely immunization coverage.
-
This study used publicly available national-level summary data published by WHO/UNICEF and did not involve personal information or human subjects research. Ethical review committee approval was not required.
HTML
| Citation: |



 Download:
Download:




