-
Introduction: On January 19, 2026, customs screening identified a foreign national arriving in Sichuan, China from Kuala Lumpur, Malaysia with an elevated body temperature. Subsequent testing yielded positive results for dengue virus (DENV) and Zika virus (ZIKV). Upon notification, local CDCs immediately launched a comprehensive epidemiological investigation, performed laboratory testing, and implemented preventive control measures.
Methods: Blood samples collected from the patient underwent quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) for viral nucleic acid detection. Next-generation sequencing (NGS) was performed on serum samples to obtain complete viral genome sequences. Phylogenetic analysis was then conducted to determine the origin and genotype of the viruses in this imported case.
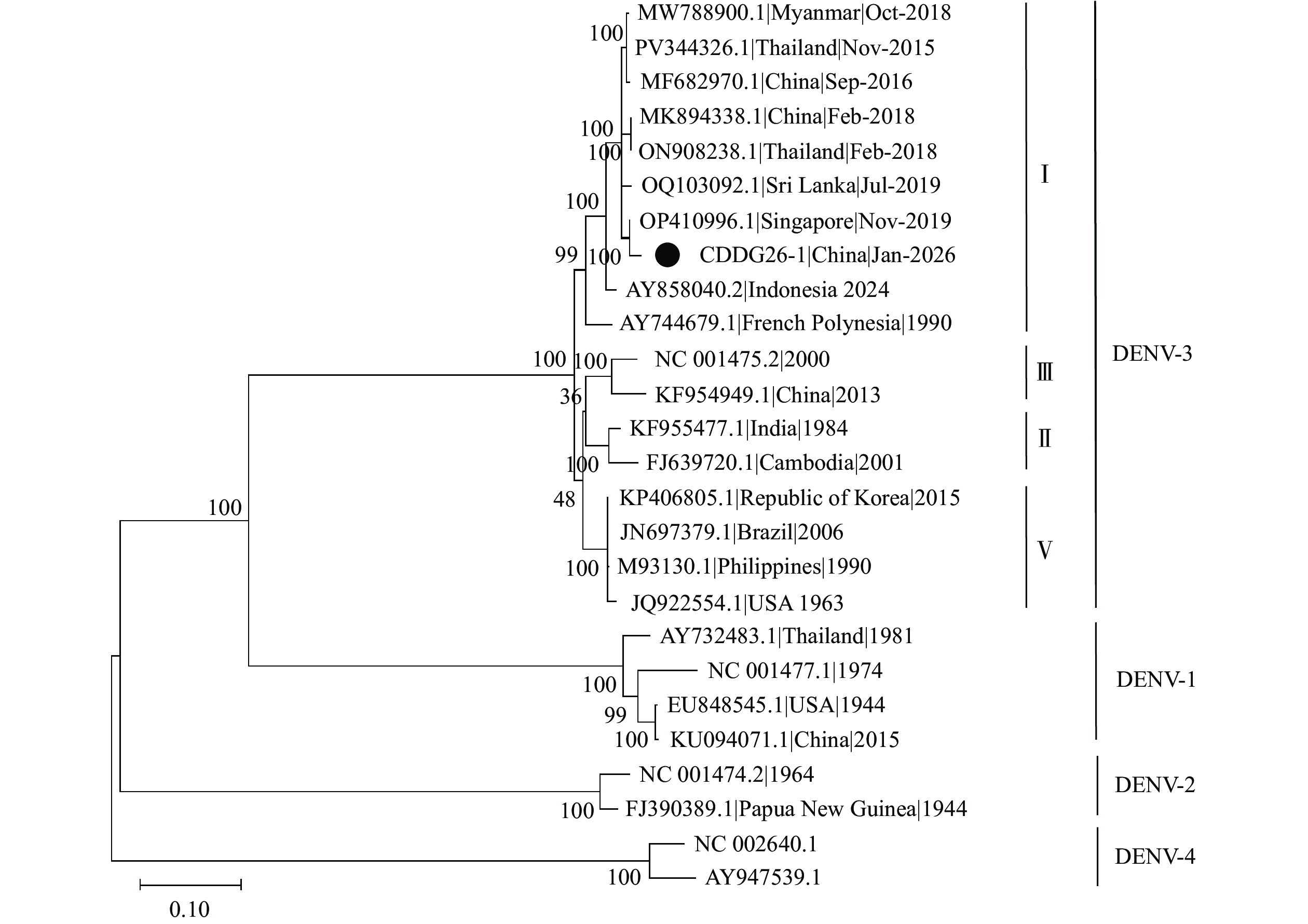
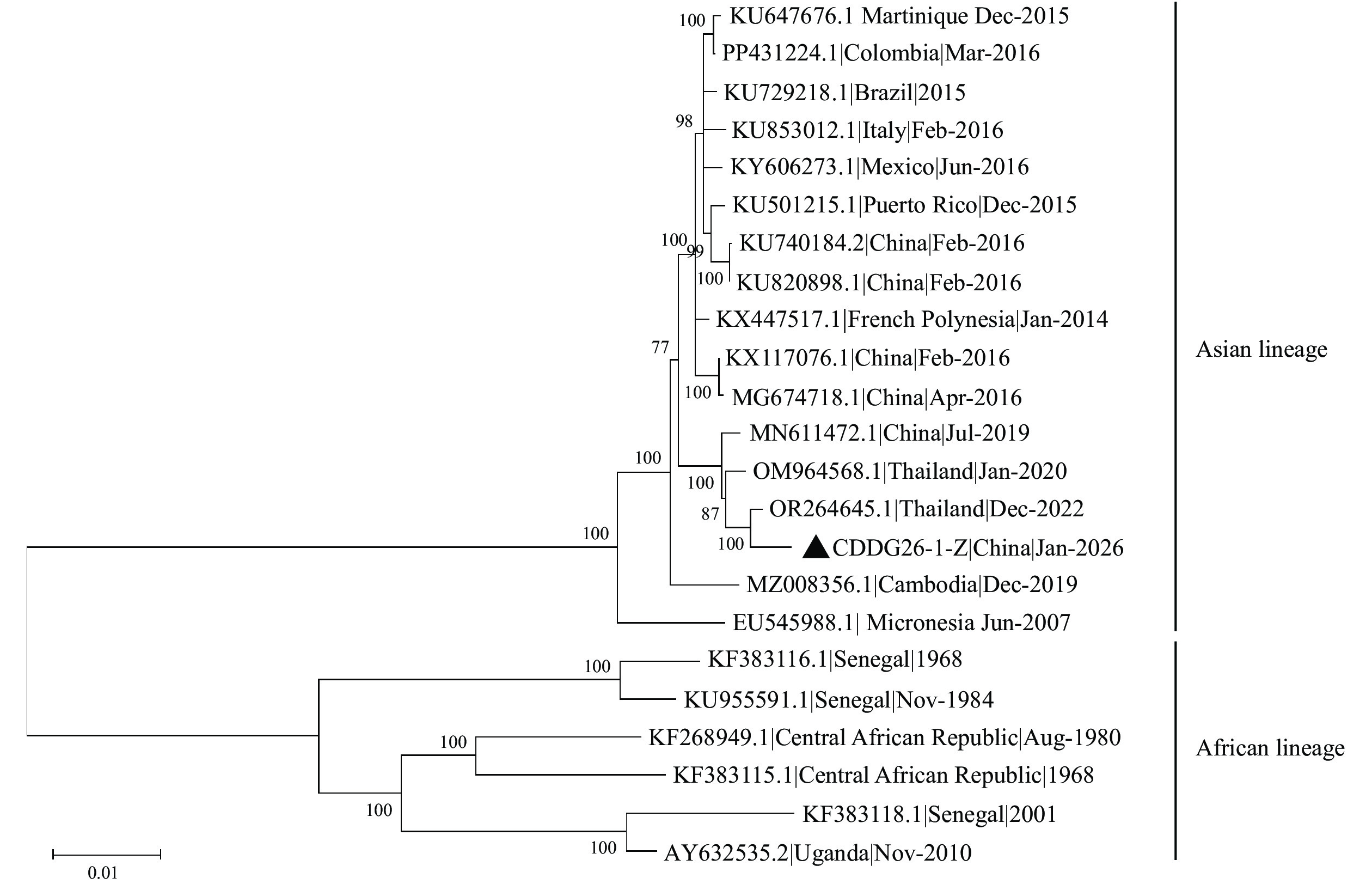
Results: The qRT-PCR analysis confirmed the simultaneous presence of DENV and ZIKV in the patient’s serum. NGS successfully yielded the complete DENV and ZIKV genome sequences. Phylogenetic analysis revealed that the ZIKV belongs to the Asian lineage, sharing 99.48% nucleotide homology with a Thailand ZIKV strain (OR264645.1); the DENV was classified as clade 3I_A.1, sharing 99.10% nucleotide homology with a Singapore DENV strain (OP410996.1).
Conclusion: Based on the epidemiological history, clinical presentation, and laboratory results, the patient was confirmed as an imported dengue/Zika coinfection case. Phylogenetic analysis demonstrated that the ZIKV and DENV strains were most closely related to those from Thailand and Singapore, respectively.
-
On January 20, 2026, the Customs notified that a Malaysian national had tested positive for dengue virus (DENV) and Zika virus (ZIKV) during port quarantine. Municipal and District CDCs promptly initiated investigation, response, and laboratory testing. On January 20 and 21, Provincial and Municipal CDCs performed laboratory analyses on the collected blood samples, with both DENV and ZIKV nucleic acid tests returning positive results. On January 23, China CDC formally confirmed the patient’s coinfection with DENV and ZIKV.
-
On January 19, 2026, a 35-year-old female Malaysian national arrived in Sichuan, China by flight from Kuala Lumpur, Malaysia. During Customs clearance, infrared temperature screening registered 37.0 ℃ (98.6 °F), and a subsequent axillary reading confirmed 37.2 °C (99.0 °F). Upon questioning by the Customs officer, she reported experiencing fever but denied rash, muscle pain, headache, conjunctival hyperemia, facial flushing, chest erythema, or neurological symptoms. The Customs collected a blood sample on-site and cleared her for passage. On January 24, she traveled by high-speed rail from Sichuan to Chongqing and flew back to Malaysia on the 25th.
The patient had been a long-term resident of Kuala Lumpur, Malaysia. Within the 14 days preceding fever onset, she denied any history of mosquito bites or blood transfusions and had not traveled to other countries or visited forested areas. Co-residing family members and colleagues reported no related symptoms. The patient traveled to China with 11 individuals from the same tour group, none of whom developed related symptoms.
On January 20, 2026, the Customs performed pathogen screening for vector-borne infectious diseases on the patient’s blood samples. Results indicated positive findings for DENV and ZIKV, prompting immediate notification to the Municipal CDC. The same day, the Municipal CDC carried out parallel testing on the samples, detecting DENV-3 and ZIKV nucleic acids. On January 21, the Provincial CDC conducted confirmatory testing with consistent results. On January 23, China CDC confirmed the positive results, and on February 24, DENV and ZIKV particles were successfully isolated and serially passaged, confirming their infectivity.
On January 22, 2026, the Municipal CDC constructed sequencing libraries from serum samples using a commercial DENV whole-genome multiplex PCR kit (MicroFuture, Beijing, China), a ZIKV whole-genome multiplex PCR kit (Macro & Micro-test, Jiangsu, China), and the Nextera XT DNA Sample Preparation and Index kit (Illumina, San Diego, CA, USA). Sequencing was performed on the Illumina MiniSeq platform, and complete DENV and ZIKV genome sequences were assembled using CLC Workbench software (version 23.0, Qiagen, Düsseldorf, Germany). Phylogenetic analysis revealed that ZIKV belongs to the Asian lineage and is genetically linked to a ZIKV strain from Thailand (OR264645.1/Dec-2022) (Figure 1), sharing 99.48% nucleotide identity. The DENV was classified as clade 3I_A.1, closely related to a DENV strain from Singapore (OP410996.1/Nov-2019) (Figure 2) with 99.10% sequence similarity.
-
Upon receiving the screening results from the Customs, Municipal and District CDCs immediately initiated coordinated prevention and control measures. Given that the patient traveled through cities with temperatures ranging from 3 °C to 9 °C, and following an assessment of local mosquito vector density and transmission risk, timely health advisories were issued to the patient and her traveling companions. Additionally, clinical experts convened a consultation for the foreign patient, with health follow-up maintained until her departure.
-
DENV and ZIKV are closely related mosquito-borne flaviviruses that share similar transmission cycles, geographic distributions, and clinical manifestations (1). According to WHO statistics, approximately half of the world’s population is now at risk of dengue (2), and while ZIKV cases declined globally from 2017 onward, transmission persists at low levels in several countries across the Americas, Asia, and Africa (3). Because they share a common vector, DENV and ZIKV endemicity may expand in concert, resulting in widespread co-circulation (4). To date, clinical studies have reported human dengue/Zika coinfections in countries such as Malaysia, Thailand, and Singapore (5–7).
In China, dengue fever cases were primarily imported during the early stages until the first local outbreak occurred in Foshan, Guangdong Province, in 1978. China’s first imported ZIKV case was confirmed in Jiangxi Province in February 2016 (8). To date, all recorded ZIKV cases in China have been imported from endemic regions, including South America, Oceania, and Southeast Asia (9). However, no imported cases of DENV and ZIKV coinfection had been reported. On January 20, 2026, the Customs reported an individual testing positive for both DENV and ZIKV nucleic acids who had departed from Kuala Lumpur, Malaysia and arrived in Sichuan, China. Following laboratory testing by Municipal and Provincial CDCs and subsequent verification by China CDC, the patient was confirmed as coinfected with both DENV and ZIKV, representing the first imported case of dengue/Zika coinfection in China. Given the patient’s arrival during winter, with low ambient temperatures and minimal mosquito activity, the overall transmission risk was relatively low. However, the possibility of similar cases entering during high mosquito activity seasons or in warmer regions cannot be ruled out.
DENV and ZIKV are both single-stranded positive-sense RNA viruses with genomes of approximately 11,000 nucleotides, encoding three structural proteins and seven non-structural proteins. DENV is classified into four serotypes based on antigenic differences in the E protein. Among these, DENV serotype 3 comprises five genotypes (G-I, G-II, G-III, G-IV, and G-V). G-I predominates in Southeast Asian countries such as Malaysia, the Philippines, and Singapore (10). ZIKV is primarily divided into African and Asian genotypes; the Asian genotype has driven global epidemics and is closely associated with neurological complications, including neonatal microcephaly and Guillain-Barré syndrome (11). Phylogenetic analysis indicated that the DENV identified in this case belongs to the 3I_A.1 branch, exhibiting the highest homology with a Singapore strain (OP410996.1); ZIKV belongs to the Asian lineage and shares high homology with reference sequence OR264645.1, originating from Thailand. However, epidemiological investigation revealed no recent travel history to Thailand or Singapore, suggesting the potential existence of a hidden transmission chain.
Dengue/Zika coinfection cases predominantly present with mild symptoms similar to those of single infections, making differential diagnosis challenging (12). Currently, China’s port-of-entry prevention and control measures for imported infectious diseases rely primarily on a “single-pathogen-targeted screening” model based on clinical symptoms and travel history, which carries a high risk of missed diagnoses and misclassification. This coinfection case underscores the need to shift port-of-entry prevention strategies toward “multi-disease co-detection and co-prevention.” On the one hand, for suspected febrile cases, clinicians should consider arbovirus coinfection based on travel history, particularly for febrile individuals arriving from regions endemic for multiple mosquito-borne diseases, to prevent serious public health risks from missed diagnoses. On the other hand, domestic efforts should prioritize developing commercial multiplex nucleic acid assay kits capable of simultaneously detecting key pathogens, including DENV, ZIKV, Chikungunya virus, yellow fever virus, Rift Valley fever virus, West Nile virus, Japanese encephalitis virus, and Plasmodium. Such a “single-sample, comprehensive screening” approach would enable source-level interruption of importation and transmission risks at points of entry. Furthermore, substantial evidence indicates that simultaneous arbovirus coinfection within mosquitoes may promote co-circulation and trigger disease outbreaks (13). Consequently, enhanced vector surveillance and control efforts are essential, alongside intensified health education campaigns to raise public awareness and ensure adherence to personal protective measures.
With global warming, accelerated urbanization, economic globalization, and increasingly frequent cross-regional travel, the prevention and control of imported vector-borne infectious diseases remains an urgent priority. The successful management of this case demonstrated highly effective collaboration among health authorities, disease control agencies, and customs departments, enabling timely detection of the imported dengue/Zika coinfection case and successful prevention of further spread. This experience highlights the need for a routine multi-departmental collaboration mechanism to facilitate real-time data sharing, establish early warning systems for infectious diseases, and strengthen overall public health security.
-
Received ethical approval from the Ethics Committee of the Chengdu Center for Disease Control and Prevention (Chengdu Institute of Health Supervision) (approval number: 2026001).
HTML
| Citation: |



 Download:
Download: