-
Oncomelania hupensis (O. hupensis) is the only intermediate host of Schistosoma japonicum (1). Guangdong Province is also a province with endemic O. hupensis, and there used to be an estimated 110 million square meters of original habitat for O. hupensis (2). In order to eliminate schistosomiasis, a team in Guangdong modified the environment to undertake O. hupensis control measures, and since 1993, live O. hupensis have disappeared in Guangdong (3). Unexpectedly, live O. hupensis were found during the spring O. hupensis survey in Qujiang District of Shaoguan City and Yingde City of Qingyuan City of Guangdong in early April of 2019 (Figure 1). In order to maintain the state of no live O. hupensis in Guangdong Province and to eliminate the risk of possible recurrence of schistosomiasis, a team in Guangdong was dispatched to carry out prevention and control measures to eliminate O. hupensis and modify its breeding area from April to August 2019 in this study.
 Figure 1.
Figure 1.The map of comprehensive management of Oncomelania hupensis in Guangdong Province in 2019.
The first O. hupensis habitat area was located in the junction area of Changjiang Dam of Shakou Town, Yingde (county-level jurisdiction), Qingyuan City, and Qunxing Village of Zhangshi Town, Qujiang District (county-level jurisdiction), Shaoguan City, which was originally an O. hupensis habitat area (Figure 1), with diverse and complex surrounding environments. The O. hupensis density survey was carried out by 5 meter systematic sampling combined with 2-frame environmental sampling. Both dead and live snails were identified, and the infection rate of Schistosoma japonicum cercariae in live snails was detected by anatomical microscope, polymerase chain reaction (PCR), and loop-mediated isothermal amplification (4). At the same time, the whole mitochondrial genome DNA of snails was sequenced and phylogenetic analysis was carried out using next generation sequencing. In order to further understand the schistosomiasis infection status of people and livestock in the affected areas, the venous blood and feces of people and cattle were collected, and schistosomiasis antibodies were detected for using indirect haemagglutination assay (IHA). The feces were detected by metacercaria incubation method and Kato Katz. In addition, in order to completely eliminate the snails, comprehensive environmental treatment was adopted in the response process. The main measures include the following: comprehensively managing ditches by weeding and clearing the surface; spraying the area with 26% metaldehyde and niclosamide suspension concentrate combined with either black ground rubber film cover or with soaking to eliminate O. hupensis; and filling old ditches and constructing new ditches and hardening three sides with smooth surfaces.
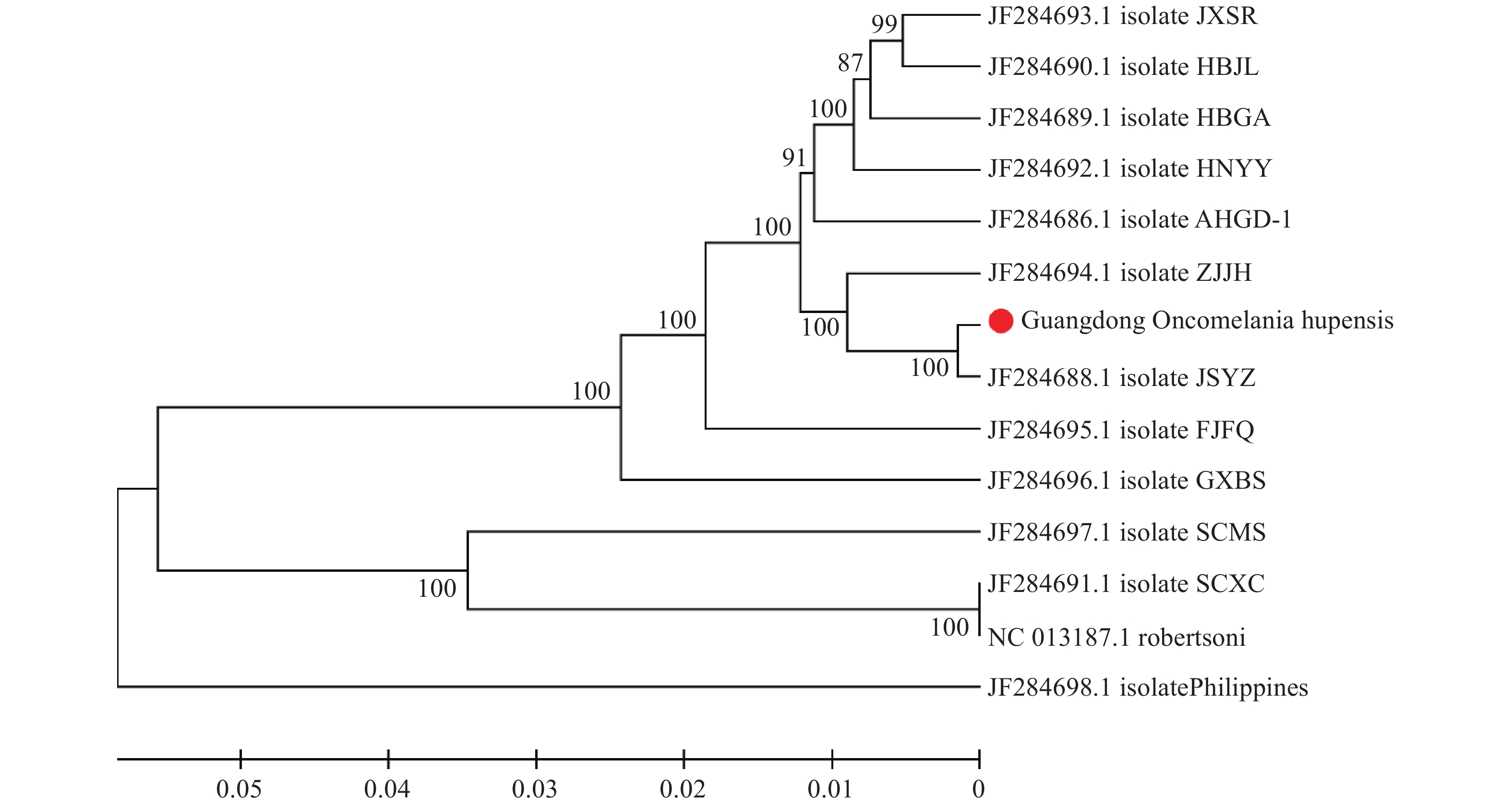
The results showed a total of 1,847 suspected environments suitable for O. hupensis habitat that were investigated, with a total area of about 2.26 million square meters. A total of 40 environments with O. hupensis were identified covering an area of about 124.7 thousand square meters. The occurrence rate of the survey frames with O. hupensis in the habitat environment was 0.32%−66.09%, and the density of live O. hupensis was 0.01−7.66/frame (Table 1). A total of 8,612 snails were collected and identified as live snails. The whole mitochondrial genome sequence (15,850 bp) of O. hupensis was obtained by second-generation deep sequencing. The results of identification and evolutionary analysis showed that O. hupensis in Guangdong Province and the Yangzhou (JF284688.1) strain in Jiangsu Province were located in the same evolutionary branch, and identification with the strain at the nucleotide level was 100% (Figure 2). A total of 4,861 human blood samples were collected, with a test response rate of more than 95%. The IHA test results were positive for schistosomiasis antibody in 14 people (0.29%), and the stool samples of 14 IHA positive people were negative for schistosomiasis eggs and metacercaria. The blood and fecal samples of 269 buffalo in all affected areas were collected, and the examination rate was 100%. No Schistosoma antibodies, eggs, and cercariae were found. In the process of environmental treatment, a total of 51.94 km of canals were newly built and repaired, 3 screw basins were newly built, 27 culverts were rebuilt, and more than 3,000 acres of farmland remediation and rehabilitation were completed. The cumulative drug snail extermination area was 1.45 million square meters. From December 2020 to May 2021, 40 environments with O. hupensis and the surrounding environments were monitored and evaluated several times, and no more live O. hupensis were detected.
County-level jurisdiction No. of snail environment Environment type Environmental area (m2) No. of survey system frames No. of live snail frames Rate of live snail frames No. of live snails collected Live Oncomelania density
(No./ frame)Qujiang 1 Ditch 78 43 6 13.95 147 3.42 Qujiang 2 Ditch 1,575 12 1 8.33 1 0.08 Qujiang 3 Ditch 400 23 5 21.74 11 0.48 Qujiang 4 Ditch 800 174 115 66.09 794 4.56 Qujiang 5 Ditch 60 26 1 3.85 1 0.04 Qujiang 6 Waste land 865 66 1 1.52 1 0.02 Qujiang 7 Waste land 5,004 133 2 1.50 2 0.02 Qujiang 8 Ditch 50 13 2 15.38 5 0.38 Qujiang 9 Ditch 50 12 4 33.33 20 1.67 Qujiang 10 Ditch 320 308 1 0.32 17 0.06 Qujiang 11 Ditch 65 28 4 14.29 16 0.57 Qujiang 12 Ditch 175 33 3 9.09 6 0.18 Qujiang 13 Waste land 2,200 262 4 1.53 16 0.06 Qujiang 14 Ditch 240 83 1 1.20 1 0.01 Qujiang 15 Waste land 30 11 3 27.27 15 1.36 Qujiang 16 Waste land 50 11 2 18.18 15 1.36 Qujiang 17 Waste land 7,178 414 54 13.04 97 0.23 Yingde 18 Waste land 10,525 32 11 34.38 164 5.13 Yingde 19 Ditch 1,592 31 8 25.81 63 2.03 Yingde 20 Waste land 22,056 55 17 30.91 179 3.25 Yingde 21 Waste land 2,653 111 26 23.42 144 1.30 Yingde 22 Waste land 1,781 70 25 35.17 154 2.20 Yingde 23 Waste land 2,164 77 7 9.09 590 7.66 Yingde 24 Waste land 7,676 77 12 15.58 28 0.36 Yingde 25 Waste land 13,603 98 21 21.43 300 3.06 Yingde 26 Waste land 8,935 36 11 30.56 28 0.78 Yingde 27 Waste land 2,175 73 5 6.85 25 0.34 Yingde 28 Ditch 4,064 138 11 7.97 37 0.27 Yingde 29 Waste land 1,752 17 2 11.76 13 0.76 Yingde 30 Waste land 337 18 8 44.44 90 5.00 Yingde 31 Ditch 259 99 42 42.42 629 6.35 Yingde 32 Waste land 10,599 347 18 5.19 89 0.26 Yingde 33 Waste land 6,215 147 4 2.72 33 0.22 Yingde 34 Waste land 6,245 162 10 6.17 189 1.17 Yingde 35 Ditch 278 175 102 58.29 726 4.15 Yingde 36 Ditch 250 43 8 18.60 25 0.58 Yingde 37 Ditch 620 169 1 0.59 1 0.01 Yingde 38 Ditch 1,370 238 44 18.49 1,312 5.51 Yingde 39 Ditch 200 35 20 57.14 76 2.17 Yingde 40 Ditch 300 107 1 0.93 1 0.01 Total 124,789 4,007 623 15.55 6,061 1.51 Table 1. Investigation on Oncomelania hupensis and its breeding area in the affected areas of Qujiang and Yingde, Guangdong Province from April to August 2019.
-
Schistosomiasis epidemic areas in Guangdong Province were mainly distributed in the middle and lower reaches of the Beijiang River in the Pearl River System (2). Guangdong Province announced the elimination of schistosomiasis
in 1985 as there were no O. hupensis found in 7 years. Since then, in 1992, an outbreak of O. hupensis reappeared in Caozhai Village, Sanshui District, Foshan City. It was found that there were 2000 m2 of O. hupensis and 3 persons with positive fecal eggs, including 1 case of previous infection and 2 cases of new infection (5). No O. hupensis had been reported in affected area in Yingde since 1982, and no sick cattle have been found since 1983 (5). Fortunately, we did not find any human and animal infections, so we inferred that the possible reasons for the recurrence of O. hupensis were as follows. First, there might be some remaining O. hupensis still living in the local environments at that time, and the environmental conditions were suitable for the reproduction of O. hupensis, which reached a certain density after many years. Second, the environment was low-lying and waterlogged during the flood season, resulting in abandoned fields and overgrown weeds, forming an environment suitable for O. hupensis habitation. Third, the environmental terrain and its altitude, the mode of production and grazing, etc., were all possible factors causing O. hupensis to spread in a large area in this region. Fourth, the local reporting of suspicious environment suitable for O. hupensis habitat was not comprehensive enough, which might have resulted in insufficient monitoring coverage and missed investigation for many years (6). In 2018, Yingde strengthened the reporting efforts and monitored the environment of the newly built datasets in 2018, subsequently finding the O. hupensis habitats. Of the 40 areas affected by O. hupensis, half were distributed along the water system, and the other half were distributed at independent points. It is analyzed that the habitat and diffusion of the remaining snails in this area might be categorized in three passive diffusion modes (7): 1) carried and spread by water flow along the river system, flooding, and waterlogging water; 2) carried by human production and living activities; and 3) carried by activity of grazing animals in affected areas. The following points were learned and implemented in this emergency response: 1) the monitoring system must be maintained and be effective by adhering to surveillance procedures, team training, and quality evaluation every year; 2) the reoccurrence of the O. hupensis epidemic was confirmed quickly, and the provincial, municipal and county levels immediately launched emergency response, quickly established multidepartment joint management mechanisms, and took emergency implementation measures; 3) according to the characteristics of the environments with O. hupensis, molluscacide was utilized with environmental reforms to eliminate O. hupensis rapidly and consolidate the effects of O. hupensis eradication; 4) environments containing O. hupensis, surrounding water systems, and suspected environments must be comprehensively managed to reduce suspected environments suitable for O. hupensis breeding and minimize the risk of O. hupensis breeding and diffusion in the affected places, and relevant departments must be synchronized and collaborate on their work.
There are some limitations in the snail disposal. Firstly, the limitation of snail traceability. At present, there is no whole genome sequence of oncomelania, so it can only be compared according to the mitochondrial genome sequence. Secondly, the limitation of snail distribution survey. Based on the current method of snail investigation, probability of missing inspection is high in the environment with low density; There may also be very few residual snails in the environment after snail disposal.
Strengthening surveillance is an important measure to consolidate and eliminate schistosomiasis. In order to consolidate the O. hupensis control effect, it is necessary to further strengthen the effect evaluation and monitoring, improve the environmental management, and conduct sustainable environmental management. In schistosomiasis surveillance areas, it is necessary to further strengthen the comprehensive reporting and registration of suspected environments suitable for O. hupensis habitation, strive to fully grasp the distribution and quantity of suspicious habitat environments, simultaneously carry out dynamic filing management, carry out rational O. hupensis monitoring, ensure monitoring coverage and O. hupensis survey quality, and prevent missing investigations.
HTML
| Citation: |



 Download:
Download: