-
Plague is a zoonotic disease caused by Yersinia pestis (1–2). Its natural cycle relies primarily on rodents and fleas, and it can infect humans under specific conditions (1,3). China is one of the countries with the most diverse types and widespread distribution of natural plague foci, encompassing various ecological zones such as alpine grasslands, desert steppes, mountainous areas, and agro-pastoral ecotones (4). Since the 1980s, China has implemented a multilevel nationwide surveillance system and maintained a high level of preparedness for rapid epidemic responses (5). However, recent factors, such as ecological restoration, climate change, shifting human activity, evolving epidemic trends, and changing ecological characteristics of foci, have introduced new dynamics. These changes pose greater challenges to existing prevention and control systems (6). By integrating national surveillance data from 2010 to 2024, this study provides a comprehensive characterization of 15 years of plague epidemiology, focus evolution, and diagnostic technology use in China, with the goal of informing evidence-based control strategies.
-
This study used surveillance data from the National Plague Prevention and Control Management Information System for 2010–2024. Descriptive epidemiological methods analyzed the dynamics of natural foci and human and animal epidemics in China. All surveillance activities, case definitions, laboratory methods, and data reporting strictly followed the National Plague Surveillance Program (2024 Revision) (5). Focus data covered spatial distribution and area changes in affected counties. Surveillance included fixed and mobile sites across 24 provincial-level administrative divisions (PLADs) and the Xinjiang Production and Construction Corps (XPCC), covering various ecological types such as alpine grasslands, desert steppes, mountainous areas, and agro-pastoral ecotones. Human case data included clinical classification, outcomes, exposure history, and epidemiological information. Case definition and classification followed the Diagnostic Criteria for Plague (WS 279-2008) (7). Animal epidemic data included pathogenic, immunological, and molecular test results for host animals and vectors. To evaluate surveillance effectiveness, annual positive rates (positive sample numbers / total tested ×100%) was calculated for various testing methods. Data organization and statistical analyses were performed using Microsoft Excel 2019 (Microsoft Corporation, Redmond, Washington, USA).
-
China has currently identified 12 types of natural foci. Between 2010 and 2024, 19 counties with new plague epidemic foci were identified. These are primarily located in Xizang, Sichuan, Xinjiang, Inner Mongolia and Yunnan PLADs. The Marmota himalayana focus (predominantly alpine habitat) showed the highest increase, with 13 new counties (13/19, 68.4%; 9 in Xizang, 4 in Sichuan). The Rhombomys opimus plague focus added two counties (10.5%), while the Meriones unguiculatus, Marmota baibacina-Spermophilus undulatus, Marmota caudata, and the Apodemus chevrieri-Eothenomys miletus foci added one (5.3%). These findings indicated that spatial expansion and belt-like intensification of natural plague foci in China are primarily concentrated in the high-altitude Xizang Plateau.
-
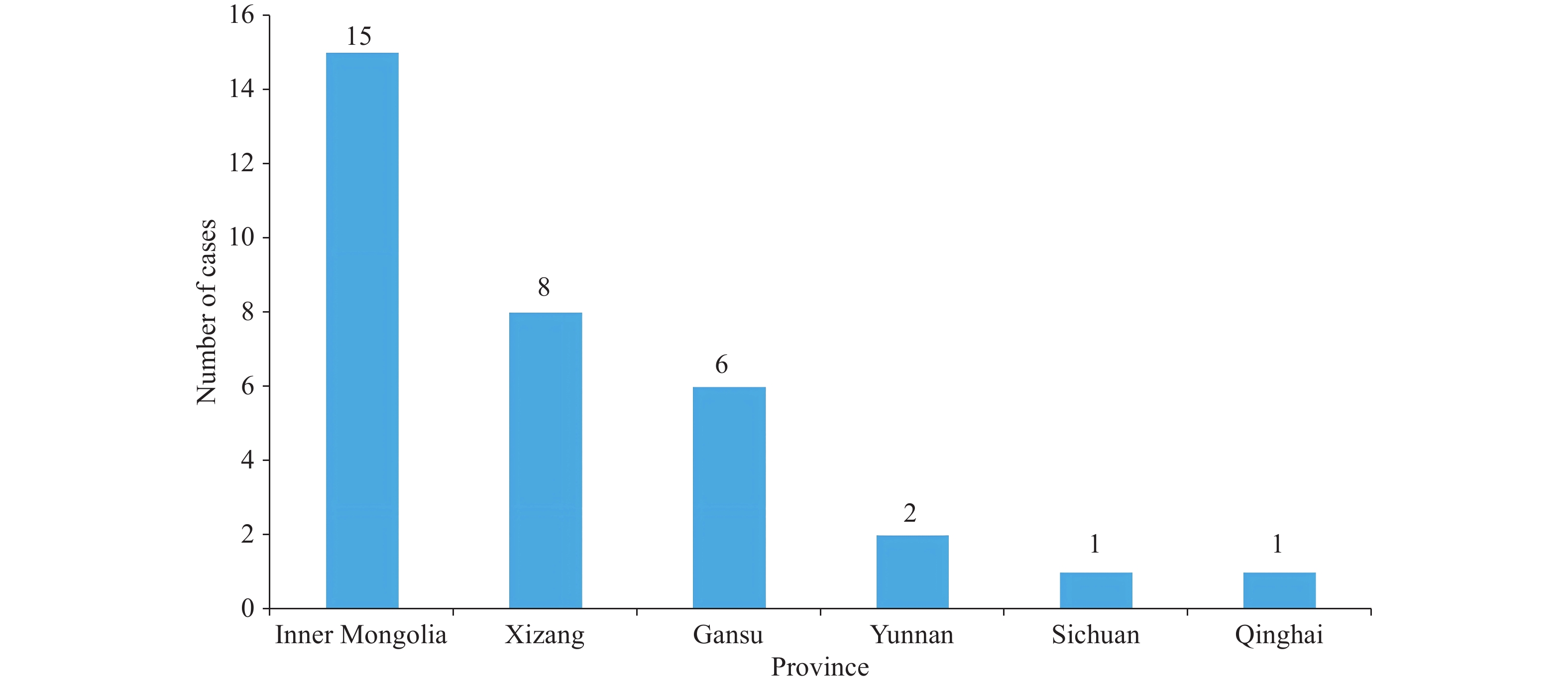
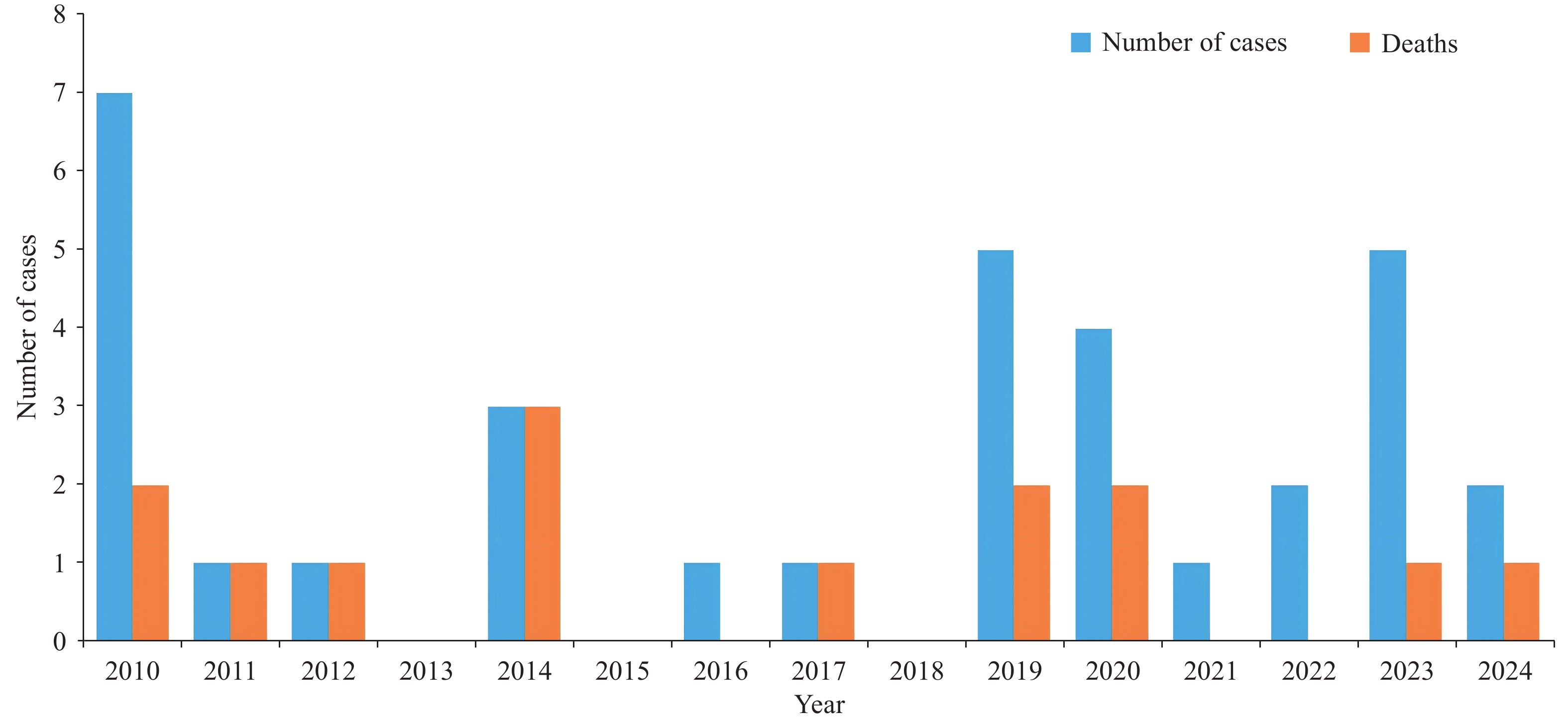
Between 2010 and 2024, 25 indigenous human plague outbreaks were reported nationwide, involving 33 cases, with 19 recoveries and 14 deaths (Figure 1). Geographically, cases were concentrated in Inner Mongolia (15, 45.5%), Xizang (8, 24.2%), and Gansu (6, 18.2%; Figure 2) PLADs. By focus type, cases originated primarily from the Marmota himalayana (16, 48.5%), Mongolian gerbil (15, 45.5%), and Asian house rat foci (2, 6.1%). Pneumonic plague was predominant (including secondary plague; 15, 45.5%), followed by bubonic (10, 30.3%), septicemic (including secondary septicemic plague; 7, 21.2%), and intestinal plague (1, 3.0%). Pneumonic plague was responsible for intrahousehold transmission and clustered outbreaks, as observed in Lang County (2010) and Sonid Right Banner (2023). Cases were predominantly male (24, 72.7%), with 9 females (27.3%). Age ranged from 3 years to 70 years, concentrated in the 20–59 age group (28, 84.8%), primarily young and middle-aged laborers. Occupational distribution was highly concentrated among herders and related occupations (27, 81.8%). The routes of infection included direct skinning or contact with infected animals, occupational exposure within plague foci, household transmission, and imported cases or cross-regional healthcare-seeking.
-
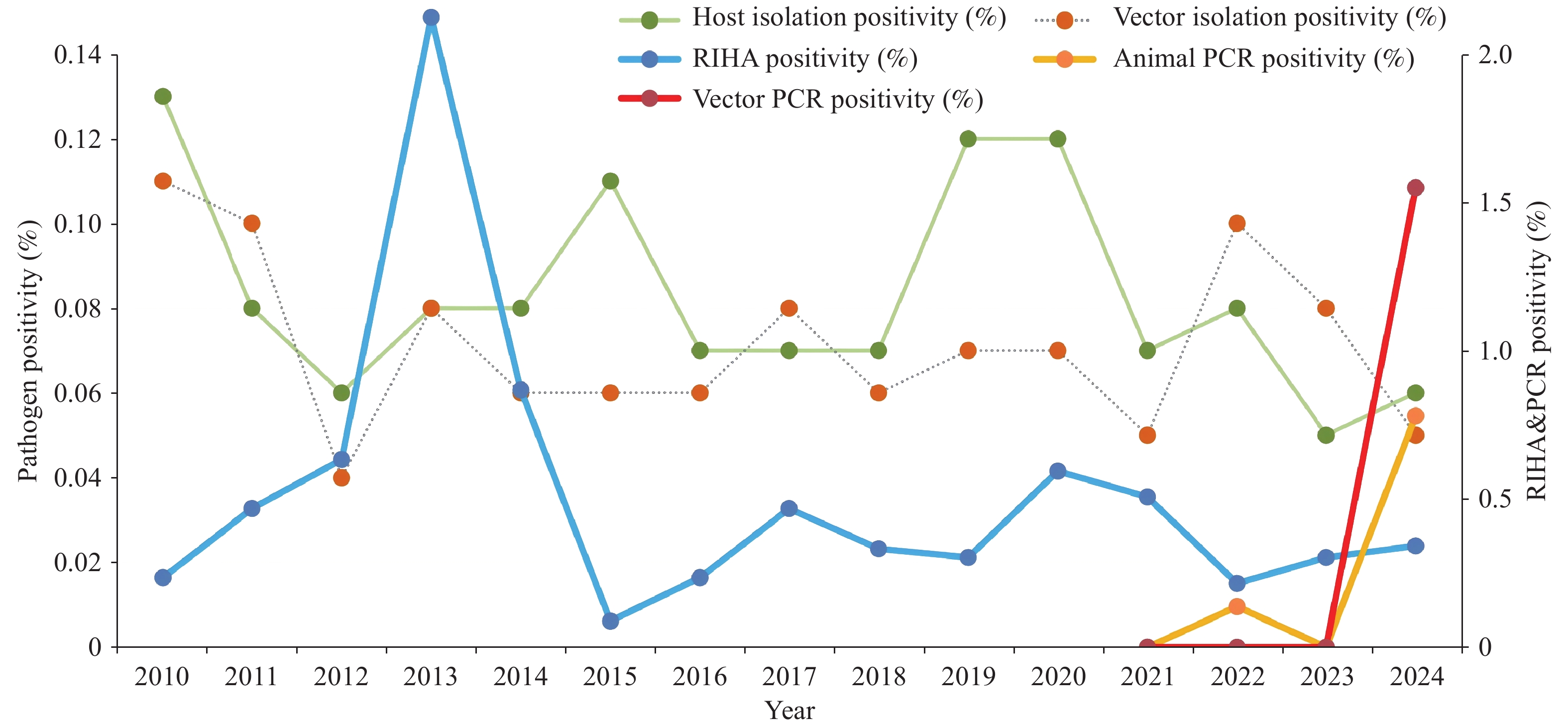
Animal plague epidemics persisted nationwide from 2010 to 2024, with significant variations in intensity across different foci. In the M. himalayana focus, pathogen isolation positivity rates were approximately 1.07% in host animals (957 positive strains) and 0.54% in vector insects (161 positive pools). In Marmota baibacina and Spermophilus undulatus, rates were approximately 0.20% in hosts (107 positive strains) and 0.16% in vectors (89 positive pools), indicating comparable sustained activity. In the Meriones unguiculatus focus, rates were approximately 0.16% in hosts (377 positive strains) and 0.19% in vectors (137 positive pools). A notable annual peak occurred in 2019, with a host animal positivity rate of approximately 0.52% (96 positive strains). Since then, animal epidemics have shown a developmental trend from east to west and north to south, with human cases reported annually for 6 consecutive years. Animal plaque has been continuously reported since the discovery of the Microtus fuscus focus. The Rhombomys opimus focus in the Junggar Basin exhibited a consecutive annual outbreak pattern, while sporadic epidemics occurred in the Apodemus chevrieri-Eothenomys miletus focus. In contrast, foci involving the Spermophilus dauricus and Marmota sibirica were quiescent. However, even in quiescent foci, sporadic positive detection underscores the need for continuous active surveillance.
Methodological analysis showed that pathogen isolation positive rates remained low over the long term. Host isolation positive rates ranged from 0.05% (2023) to 0.13% (2010), and vector rates from 0.04% (2012) to 0.11% (2010). For immunological detection, the indirect hemagglutination assay (IHA) positivity rate ranged from 0.08% (2016–2022) to 0.25% (2013). The reverse indirect hemagglutination assay (RIHA) positivity rate remained mostly below 1%, although it reached 2.18% in 2013. In 2024, animal and insect PCR-positive rates were 0.80% and 1.59%, respectively, both significantly higher than pathogen isolation results during the same period (Figure 3).
-
Ecological Factors: The northward shift of isohyets (8) and the “warming and wetting” trend (9–11) have expanded the ecological space for host animals, driving the expansion of plague foci along climatic-ecological transition zones, a mechanism supported by the spatial clustering of newly affected counties.
Enhanced Surveillance Capability: The systematic strengthening of surveillance network technology and coverage between 2010 and 2024 has significantly improved the sensitivity of identifying potential risk areas.
-
Surveillance data from 2010 to 2024 indicated that human plague in China remained at a low level of sporadic occurrence. Cases were mostly sporadic and primarily involved “first-generation cases” resulting from direct contact (hunting, skinning, eating, and biting). Existing prevention and control systems have proven effective in blocking human-to-human transmission.
Geographically, human cases were concentrated in Inner Mongolia, Xizang, and Gansu (>80% of cases). By focus type, cases were predominantly distributed in the Marmota himalayana (48.5%) and Meriones unguiculatus (45.5%) foci, confirming the overlap between high-risk geographic areas and specific host foci. Furthermore, outbreaks in new areas resulting from dynamic changes in plague foci, such as the 2012 Litang County incident in Sichuan, highlight the potential risks posed by marginal zones and climatic anomalies (12–13).
Between 2010 and 2024, multiple cross-regional transmission events occurred in China, typified by the 2019 imported case from Inner Mongolia to Beijing (14). This underscores the potential risk of plague spreading from traditionally endemic areas to densely populated metropolitan regions. Therefore, the comprehensive measures encapsulated in the “Five Earlies and One Localization” principle, early detection, early reporting, early diagnosis, early isolation, early treatment, and localization of treatment, must be strictly implemented to rapidly interrupt transmission chains and control outbreaks.
-
Surveillance data from 2010 to 2024 indicate that animal epidemics are highly spatially concentrated, primarily distributed in active foci involving species such as Marmota himalayana, Marmota baibacina, Spermophilus undulatus, and Meriones unguiculatus. The active periods of animal epidemics in these regions demonstrate spatiotemporal consistency with human case reports, suggesting that animal epidemics could serve as leading indicators of human plague risk. In terms of detection methods, the positivity rate of traditional pathogen isolation remains consistently low, while immunological assays yield persistently positive results, indicating widespread previous and latent infections among host animals. Given the large host population and complex host-vector network, once host density or climatic conditions exceed a certain threshold, dormant plague foci may be reactivated (15). Therefore, long-term systematic surveillance should be conducted in key areas (16–17). No single technology can simultaneously achieve speed, sensitivity, and confirmation. Therefore, a technological portfolio of “rapid PCR screening, immunological interpretation, and pathogen isolation confirmation” is recommended, supported by standardized quality control to ensure cross-regional comparability.
-
The current situation for plague prevention and control in China is complex, and the potential public health threats it poses cannot be ignored. To address these challenges, the prevention and control system can be optimized based on the following aspects:
Surveillance Precision: Identify core risk areas to enable precise resource allocation; Early Warning Technology: Build intelligent early warning models using multi-source data to enhance the capability for early epidemic identification; Prevention and Control Focus: Continuously strengthen basic prevention and control capacity in high-incidence areas, and emergency response capabilities for plague; Pre-positioned Resources: Proactively pre-position medical supplies and strengthen specialized personnel training before the epidemic season to reduce outbreak response start-up time; Localized Treatment: Establish a “treatment on-site” support system to reduce the risk of epidemic spread caused by cross-regional medical seeking.
Sample Size Limitations: Only 33 human cases were reported nationwide during the study period; Spatial Heterogeneity of the Surveillance Network: Surveillance site distribution may be uneven due to practical factors, including historical disease activity, geographical accessibility, and prevention and control resource allocation; Feasibility of Technology Promotion: High cost and stringent requirements of PCR and molecular techniques may limit their standardized application at the grassroots level.
-
From an international research perspective, plague has stable natural focus distributions in a few countries and regions across the Americas, Asia, and Africa (1,18). International studies have indicated that ecological factors, such as climate warming, changes in precipitation patterns, and vegetation “greening,” can promote the activation and spatial expansion of these foci by influencing host and vector population dynamics (16,19). This study found that newly identified or marginally active foci in China were predominantly distributed along climate-ecological transition zones, a finding consistent with international research. In terms of surveillance and prevention, the integration of molecular detection techniques, such as PCR, with molecular typing and molecular tracing methods has become an international trend (20–21).
Surveillance data from 2010 to 2024 indicate that the plague situation in China remains complex, characterized by “sporadic human cases, active animal epidemics in some foci, and slow expansion of natural foci.” To address these challenges, future prevention and control efforts should establish a tiered technological system of "PCR rapid screening, immunological trend analysis, and pathogen isolation for confirmation and traceability." This should be coupled with strengthened cross-provincial/regional collaboration and rapid data sharing, thereby systematically enhancing early warning and enabling precise responses, and continuously consolidating prevention and control achievements.
HTML
Distribution of Natural Plague Foci
Characteristics of Human Plague
Characteristics of Animal Plague
Analysis of Factors for Natural Plague Foci Expansion
Analysis of Human Plague Epidemiological Characteristics
Animal Plague and Surveillance Technologies
Risk Identification and Control Measures
International Comparison and Implications for Prevention and Control Strategies
| Citation: |



 Download:
Download: