-
In January 2026, a 53-year-old male migrant worker was diagnosed with fatal rabies in Zhejiang Province, China. The patient had a documented 10-year history of human immunodeficiency virus (HIV) infection and was receiving regular antiretroviral therapy with Biktarvy. At the time of hospital admission, laboratory testing on January 12 demonstrated effective virological control, with a CD4+ T-lymphocyte count of 335 cells/μL and an undetectable HIV-1 viral load; however, syphilis serology was positive. The patient had recently returned to Jinhua City, Zhejiang Province, from Tongren City, Guizhou Province, in December 2025.
Approximately 4 months prior to symptom onset, on September 10, 2025, the patient sustained a bleeding finger bite from his domestic dog while residing in Songtao County, Guizhou — a known rabies-endemic area (1). Critically, no post-exposure prophylaxis (PEP) was administered following this exposure (2). The dog was slaughtered and consumed shortly after the incident. On January 8, 2026, the patient developed influenza-like symptoms that rapidly progressed. By January 10, symptoms had evolved to include vertigo and limb weakness. Given his immunocompromised status, initial differential diagnoses focused on neurosyphilis and HIV-associated opportunistic infections. Targeted next-generation sequencing (tNGS) of cerebrospinal fluid (CSF), performed by Hangzhou Adicon Clinical Laboratories, failed to identify a pathogen.
On January 16, the emergence of pathognomonic rabies symptoms — hypersalivation and hydrophobia — prompted urgent reassessment. Unbiased metagenomic next-generation sequencing (mNGS) of the CSF, also conducted by Hangzhou Adicon Clinical Laboratories, successfully identified rabies virus (RABV) RNA, establishing a definitive diagnosis of furious rabies. The patient died on January 17 (Figure 1). This case illustrates how concurrent HIV infection and syphilis can obscure the early clinical recognition of rabies, while demonstrating the critical diagnostic value of mNGS in resolving complex, atypical presentations of infectious encephalitis.
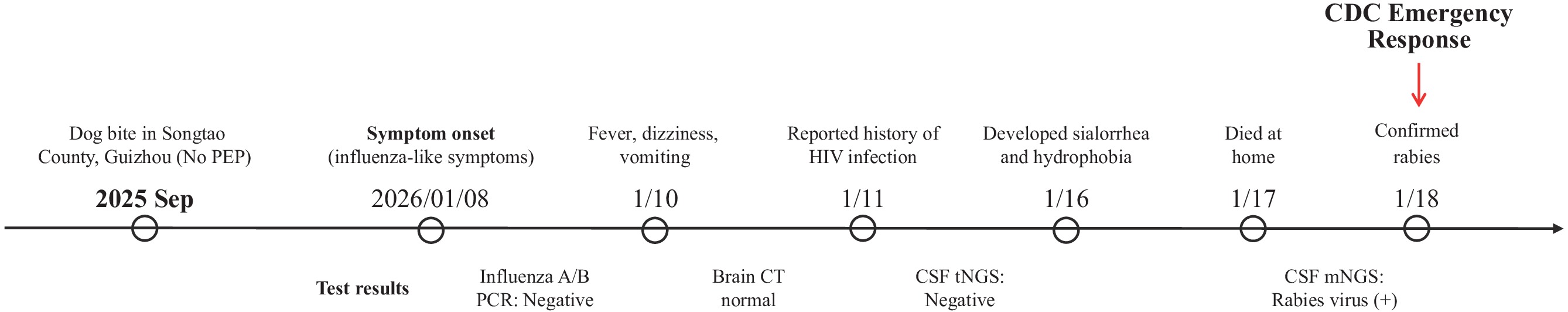 Figure 1.
Figure 1.Timeline of exposure, symptom onset, diagnostic evaluation, and clinical outcome in a fatal human rabies case with HIV co-infection, Zhejiang Province, China.
Abbreviation: CSF=cerebrospinal fluid; CT=computed tomography; HIV=human immunodeficiency virus; mNGS=metagenomic next-generation sequencing; PCR=polymerase chain reaction; tNGS=targeted next-generation sequencing; PEP=post-exposure prophylaxis.Upon laboratory confirmation, the Jinhua CDC initiated an epidemiological investigation that identified 23 close contacts. Contact classification included family caregivers (n=6), healthcare workers (n=10), and community members (n=7; comprising five individuals who consumed the dog meat, one funeral handler, and one sanitation worker). All contacts underwent standardized risk assessment and received PEP in accordance with national rabies prevention guidelines.
Because the exposure event occurred retrospectively in Guizhou Province, the precise number of villagers participating in the dog slaughtering could not be definitively established. All identified high-risk individuals were nevertheless counseled to initiate PEP. A cross-provincial notification was transmitted to the Guizhou CDC to enable coordinated source investigation and contact tracing efforts. As of February 2026, no secondary transmission has been documented. All close contacts remain under active surveillance for a minimum of 6 months, consistent with national rabies prevention protocols.
This case demonstrates critical challenges in diagnosing rabies when concurrent conditions complicate clinical presentation. First, clinicians evaluating patients with acute neurological symptoms should systematically elicit detailed animal exposure histories, particularly in individuals with immunocompromising conditions such as HIV infection. Co-morbidities can mask pathognomonic rabies features, delaying recognition even in patients with well-controlled viral loads (3-4). Second, when conventional diagnostic approaches fail to identify causative pathogens in unexplained encephalitis, unbiased mNGS provides a powerful tool for detecting rabies virus and other atypical neurotropic pathogens (5). Third, rabies-endemic rural areas require strengthened public health infrastructure to ensure timely PEP access and effective risk communication campaigns that explicitly discourage slaughtering and consuming animals suspected of rabies infection. Finally, protecting migrant worker populations demands coordinated interprovincial surveillance systems, standardized contact tracing protocols, and targeted health education initiatives implemented in both labor-exporting and labor-receiving regions to address the unique vulnerabilities of mobile populations.
HTML
| Citation: |



 Download:
Download:




