-
The National Health Commission of the People’s Republic of China and the State Administration for Market Regulation have issued the National Food Safety Standard (GB2762-2022), which delineates the maximum limits (ML) of contaminants in food. This standard will be implemented on June 30, 2023. It currently maintains the ML of cadmium in rice (including unhusked rice, husked rice, polished rice) at 0.2 mg/kg, a value first established 40 years ago in GBn238-1984.
Considering the higher ML of 0.4 mg/kg outlined by the Codex Alimentarius Commission (CAC) and the lower limit of 0.15 mg/kg recommended by the European Food Safety Authority’s (EFSA) Panel on Contaminants in the Food Chain (CONTAM Panel), a review of the Chinese standard was deemed necessary. The primary objective was to determine whether the current provisional tolerable monthly intake (PTMI) of 25 μg/kg body weight (b.w.) for cadmium, as established by the Joint FAO/WHO Expert Committee on Food Additives (JECFA), remains appropriate for China.
To reach our recommendation, we considered additional data on the dietary consumption patterns and corresponding biomarkers of exposure for the Chinese population. We also conducted an updated literature review and examined assessments performed by both JECFA and the EFSA CONTAM Panel. Based on these findings, we recommend maintaining the PTMI of cadmium exposure in China at 25 μg/kg b.w. This recommendation provides a scientific foundation for the newly issued ML of cadmium in rice.
-
Cadmium is classified as a type I carcinogen by the International Agency for Research on Cancer (IARC) (1), and exposure has been associated with a range of cancers. Long-term exposure to cadmium primarily exerts toxic effects on the kidneys, but also affects the bones (2). Food is the major source of cadmium exposure for the non-smoking general population, contributing up to 90% of the total human cadmium intake (3-4). Rice is more susceptible to cadmium contamination than other crops (5). Furthermore, China is the largest rice-producing and consuming country globally, with rice production accounting for over one-third of the total domestic grain output. Therefore, monitoring cadmium ML in rice is not only a public health measure; it is also part of the nationwide surveillance and control efforts focused on the quality of agricultural products. These practices protect domestic rice traders and contribute to maintaining the safety of food sources in the Chinese mainland.
The ML of cadmium in food was initially established in China in 1984 (GBn238-1984) and has undergone multiple re-evaluations (GB15201-1994, GB2762-2005, GB2762-2012, GB2762-2017), based on the provisional tolerable weekly intake (PTWI) of cadmium set forth by the JECFA (revised to PTMI in 2010). As Table 1 illustrates, the limit values for cadmium in rice range from 0.1 to 0.4 among major rice-trading countries. The ongoing debate regarding the precise allowed level remains unresolved, as evidenced by the current discrepancy between CAC and EFSA guidelines. Given the variability in consumption patterns, cadmium exposure, absorption, and metabolism among diverse populations, a universally accepted standard may not be suitable. As a result, establishing a health-based guidance value (HBGV) for dietary exposure to cadmium in the Chinese mainland is crucial to determine an acceptable cadmium limit in rice for the domestic market.
Nations/regions/
organizationsCadmium limits in rice (mg/kg) Unhusked rice/husked rice Polished rice/rice (flour) China (including Hong Kong SAR) 0.2 0.2 Codex Alimentarius Commission (CAC) −* 0.4 The European Union (EU) −* 0.15 The Republic of Korea −* 0.2 Singapore −* 0.2 Japan 0.4 0.4 Russia −* 0.1 Australia −* 0.1 New Zealand −* 0.1 Thailand −* 0.4 Vietnam −* 0.4 Abbreviation: ML=maximum limits; SAR=Special Administrative Region.
* Data unavailable due to no limit for unhusked/husked rice or undifferentiated limits for rice in those countries or organizations.Table 1. The ML of cadmium in rice in China and other major rice trading countries or organizations.
-
The research to establish the provisional HBGV for cadmium exposure in food was proposed by the National Expert Committee for Food Safety Risk Assessment, entrusted by the National Health Commission, and conducted by the Secretariat of the Expert Committee for Food Safety Risk Assessment in the China National Center for Food Safety Risk Assessment (CFSA).
The CFSA team analyzed data gathered from a representative sample of the Chinese population residing in cadmium-contaminated regions. The data included food consumption, contamination levels in food, cadmium absorption and metabolism characteristics in humans, and recently updated literature information. Renal dysfunction was considered as the primary adverse health outcome from cadmium exposure, using β2-microglobulin (B2M) as a biomarker for renal tubular effects.
A concentration-effect model was established based on the biomarker of exposure (urinary cadmium concentration) and the biomarker of response (B2M concentration) to predict the benchmark dose (BMD) or threshold of urinary cadmium as a reference point (RP). A one-compartment toxicokinetic (TK) model and a physiologically based toxicokinetic (PBTK) model specifically designed for the Chinese population were employed to correlate urinary cadmium concentration with dietary cadmium intake. Consequently, the provisional HBGV was estimated.
The study included a total of 7,152 participants from 6 provincial-level administrative divisions (PLADs) (Sichuan, Hunan, Guangdong, Jiangxi, Zhejiang, and Shanghai). To investigate the chronic toxic effects of cadmium exposure, this research focused on local residents who had consumed locally grown rice for 30 years or more. As a result, 67.0% of the sample population consisted of individuals aged 50 years and above. Food consumption data were acquired using a 24-hour recall method, during which participants provided a detailed account of their food consumption over the previous day via an interview. Morning midstream urine specimens were collected from the participants. Urinary cadmium and food cadmium levels were measured using inductively coupled plasma mass spectrometry, while urinary B2M concentration was assessed through an automatic biochemical analyzer.
-
The methodologies employed in this study closely followed those utilized by the EFSA CONTAM Panel and the JECFA (3) for deriving the HBGV for dietary cadmium. Both organizations conducted a step-wise toxicodynamic/toxicokinetic assessment (3,6-8). The two primary components of this analysis included a concentration-effect model, which correlated urinary cadmium concentrations with B2M levels, and a toxicokinetic model, which associated urinary cadmium concentrations with dietary cadmium intake. These models were executed and critically assessed to ensure accuracy and reliability in the findings.
The assessments conducted by EFSA and JECFA were based on a meta-analysis derived from a systematic review of epidemiological studies, with reported summary values of urinary cadmium and B2M concentrations. The present study evaluates both individual data from surveys conducted among Chinese residents and summary data from an updated meta-analysis to derive the final HBGV. In addition to the Hill model adopted by EFSA, the piecewise linear model and generalized additive model were also utilized to predict the RP of urinary cadmium. A one-compartment TK model and a more comprehensive PBTK model, optimized with exposure characteristics of non-smoking residents in Shanghai, were employed to derive the dietary cadmium intake from different RPs of urinary cadmium (9). Figure 1 illustrates the technical process used to derive the provisional HBGV.
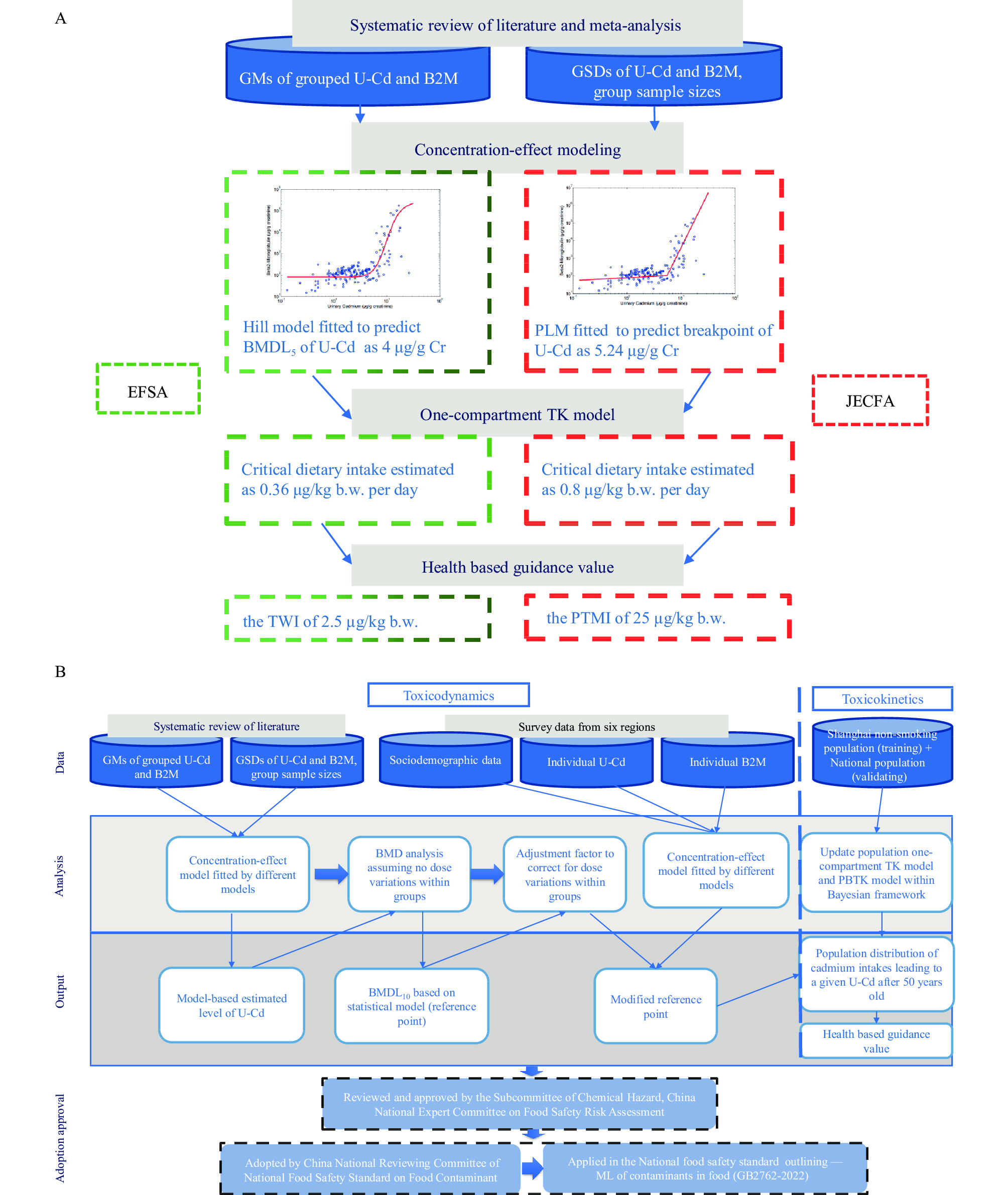 Figure 1.
Figure 1.The basis and process of establishment of the standard for the ML of cadmium in rice. (A) The process of the HBGV derivation by the EFSA CONTAM Panel and the JECFA. (B) Technical process of the HBGV derivation with procedure of approval and adoption for establishment of the standard for ML in China.
Abbreviation: ML=maximum limits; GM=geometric mean; GSD=geometric standard deviation; U-Cd=urinary cadmium; B2M=β2-microglobulin; BMDL5=benchmark dose lower confidence limit at the benchmark response of 5%; Cr=creatinine; PLM=piece-wise linear model; HBGV=health based guidance value; TWI=tolerable weekly intake; PTMI=provisional tolerable monthly intake; EFSA CONTAM Panel=European Food Safety Authority’s Panel on Contaminants in the Food Chain; JECFA=Joint FAO/WHO Expert Committee on Food Additives. -
The assessment report underwent review and approval during the second meeting of the Sub-Committee on Chemical Hazard, part of the National Food Safety Risk Assessment Expert Committee, on May 27, 2021. The results with comparison to EFSA, JECFA, and similar domestic studies (10) were listed in Table 2.
Phase outcomes for HBVG derivation BMDL5
(EFSA)BMDL5
(JECFA)BMDL5
(current study)BMDL10
(current study)Thresholds derived by generalized additive model BMDL5
(Meta-analysis)BMDL10
(Meta-analysis)BMDL10
(Ke’s study)Reference point of urinary cadmium (μg/g Cr) 4
(1)*5.24
(4.9–5.57)†2.11
(0.88)*4.46
(1.86)*0.85
(0.62–0.98)†1.70
(0.71)*1.95
(0.81)*2.0 (M)§
1.69 (F)§TK model TDI [μg/(kg b.w.·day)] 0.36 1.2
(0.8–1.8) †0.55 1.17 0.54
(0.39–0.62) †0.48 0.52 1.25 (M)
1.06 (F)PTMI [μg/(kg b.w.·month)] 10.8 25 16.5 35.1 16.2
(11.7–18.6) †14.4 15.6 37.5 (M)
31.8 (F)PBTK model TDI [μg/(kg b.w.·day)] −¶ −¶ 0.37 0.95 0.36
(0.23–0.41) †0.30 0.35 1.04 (M)
0.84 (F)PTMI [μg/(kg b.w.·month)] −¶ −¶ 11.1 28.5 10.8
(6.9–12.3) †9.0 10.5 31.2 (M)
25.2 (F)Abbreviations: TDI=tolerable daily intake; PTMI=provisional tolerable monthly intake; EFSA CONTAM Panel=European Food Safety Authority’s Panel on Contaminants in the Food Chain; JECFA=Joint FAO/WHO Expert Committee on Food Additives; HBGV=health based guidance value; BMDL5=benchmark dose lower confidence limit at the benchmark response of 5%; BMDL10=benchmark dose lower confidence limit at the benchmark response of 10%; Cr=creatinine; TK=toxicokinetic; PBTK=physiologically based toxicokinetic.
* Values in brackets have been adjusted for the uncertainty factor.
§ M: Males; F: Females.
† 95% confidence interval.
¶ Data unavailable.Table 2. The estimated TDI and PTMI by the current study, the EFSA CONTAM Panel, the JECFA and other recent studies.
The estimated RPs for urinary cadmium in the Chinese population range from 0.71 to 1.86 μg/g creatinine (Cr), with PTMI values of 14.4 to 35.1 μg/kg b.w. based on TK model and 9.0 to 28.5 μg/kg b.w. based on the PBTK model. When considering the BMDL10 as an RP with high-dose effects alongside the conservative PBTK model, the calculated PTMI equates to 28.5 μg/kg b.w. In contrast, using the BMDL5 result in conjunction with the TK model yields a calculated PTMI of 16.5 μg/kg b.w. Given the current cadmium exposure status in China, the recommended PTMI for cadmium exposure for the Chinese population is 25 μg/kg b.w., aligning with the recommendation proposed by JECFA.
-
Based on the PTMI value derived from the Chinese population, this study assessed the health risks associated with different cadmium MLs in rice among various regions and age groups in China. The assessment evaluated the level of cadmium exposure in rice and its contribution rate to total dietary exposure using consumption data from the National Food Safety Surveillance (2015–2020), China Nutrition and Health Surveillance (2015–2017), and the Chinese Total Diet Study (2012). Results indicated that the nationwide exposure level to cadmium from rice consumption was generally lower than the PTMI. However, high-consuming populations, children under six years old, and individuals residing in Southern China exhibited higher cadmium exposure levels than the PTMI threshold. Strict implementation of the current cadmium ML of 0.2 mg/kg in rice could reduce dietary exposure to cadmium by 2% to 20% for residents with high dietary cadmium exposure in four southern PLADs in the Chinese mainland. Nevertheless, the potential for a subset of these high consumers to exceed the PTMI remains, indicating that adjusting the ML value from 0.2 mg/kg to 0.4 mg/kg is not recommended.
On July 20, 2021, during the seventh meeting of the Chief Technical Officers of the China National Reviewing Committee of National Food Safety Standards, it was noted that:
1) The total dietary cadmium intake among the Chinese population was found to be near the health guidance value or exceeding the PTMI in specific regions of China. Consequently, there is no justifiable evidence to support increasing the limit in accordance with the CAC recommendation.
2) Currently, no processing methods exist to reduce cadmium levels in rice from 0.4 mg/kg to 0.2 mg/kg or lower before consumption.
3) The existing literature provides insufficient evidence regarding the risk assessment of cadmium exposure stemming from the consumption of husked rice and rice designated for food processing.
4) Globally, standards for cadmium ML in rice are becoming more stringent, as evidenced by the recommendation from the EFSA.
5) Relaxing the ML standard from 0.2 mg/kg to 0.4 mg/kg without substantial evidence may lead to increased concerns about food safety among consumers.
In conclusion, this study recommends upholding ML of 0.2 mg/kg for cadmium in rice, as established by the GB 2762-2017 guidelines.
-
No conflicts of interest.
-
Guangdong Provincial CDC, Jiangxi Provincial CDC, Hunan Provincial CDC, Guangxi Provincial CDC, West China School of Public Health, Sichuan University; and also for Shanghai Suburban Adult Cohort and Biobank (SSACB) study.
HTML
| Citation: |



 Download:
Download:




