-
Novel coronavirus pneumonia (coronavirus disease 2019, COVID-19) can infect anyone and causes symptoms ranging from mild to severe. Previous studies demonstrated that severe COVID-19 had more unfavourable treatment outcomes compared to non-severe COVID-19 (1-2). Early diagnosis and timely treatment were essential to cure severe COVID-19 patients and curb the spread of disease. Yet, rapid and convenient inflammatory markers for identifying severe COVID-19 infection have not been well studied, especially for young patients. Evidence has shown that the lymphocytes count (especially the CD4+ and CD8+ T cell counts) decreased as infection progressed (3). Neutrophils and platelets were found to be important mediators of inflammation. In severe COVID-19 cases, neutrophil counts were increased (4), and platelet accumulation was common (5). Neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) have been used to evaluate systemic inflammation in neoplastic and cardiovascular diseases (6-7). Using data from 452 confirmed COVID-19 cases, we examined whether NLR and PLR values on admission may help us identify severe patients upon admission.
To better understand the association between NLR, PLR, and severity of patients with COVID-19, we conducted a multi-center observational study in 41 hospitals from 7 provincial-level administrative divisions (PLADs) of China, i.e., Shanxi, Hebei, Heilongjiang, Shaanxi, Anhui, Guangxi, and Sichuan between January 21, 2020 and February 10, 2020 (Supplementary Table S1). The implementation sites of the 7 PLADs were selected based on the geographical distribution (namely Eastern, Western, and Central regions of China), and 41 hospitals from the 7 PLADs were chosen based on their willingness to participate. All of these hospitals were designated hospitals for treating COVID-19 patients.
In our study, all COVID-19 patients enrolled were confirmed by a laboratory test; the patients were excluded if core data such as routine blood laboratory data was incomplete at admission. Medical records of these patients were collected. The study was approved by the National Administration of Traditional Chinese Medicine and Institutional Review Board at each participating hospital. Due to the urgency in treating COVID-19 patients, the requirement for written informed consent from study participants was replaced by verbal consent. All data were supplied and analyzed in an anonymous format, without access to personal identifying information.
This study has been registered by the Chinese Clinical Trial Registry (Registration Number: ChiCTR2100042177) and approved by the Ethics Committee of the Institute of Clinical Basic Medicine of Chinese Medicine, China Academy of Chinese Medical Sciences (NO: P20009/PJ09).
De-identified demographic data [sex, age, body mass index (BMI), and comorbidity] and onset symptoms (fever, cough, dry cough, fatigue, shortness of breath, and diarrhea) were collected from patients’ medical records. Results of complete blood count upon admission — including neutrophil count, platelet count, and lymphocyte count to calculate NLR and PLR — were collected.
Patients were divided into two groups of non-severe and severe based on their physician’s clinical diagnosis after admission. Severe cases were defined as having any of the following: 1)respiratory distress; 2)pulse oxygen saturation ≤93%; or 3)arterial partial pressure of oxygen (PaO2) / oxygen concentration ≤300 mmHg.
Multivariable logistic regression models were used to estimate the odds ratio (OR) and 95% confidence interval (95% CI) between NLR and PLR and patient’s clinical severity of COVID-19. Receiver-operating characteristic (ROC) curves were used to assess the diagnostic value for identifying severe COVID-19 cases. In subgroup analyses, we stratified by sex and age (<40 years, 40–59 years, and ≥60 years).
A total of 452 patients were analyzed in our study between January 21, 2020 and February 10, 2020. The median age of patients was 45 years [interquartile range (IQR): 33.0, 57.0]; 50.9% of the participants were men; 33.8% had at least one comorbidity and the median BMI was 24.3 (IQR: 21.5, 26.4). Of 451 cases, 11.9% of severe and 4.0% of critical cases; 84.1% of non-severe cases including 41 mild cases and 339 moderate cases, respectively. The most common symptoms were cough (61.3%), fever (49.1%), and fatigue (37.6%), as seen in Table 1. The median (IQR, Q1–Q3) NLR and PLR in severe COVID-19 patients on admission were 5.4 (3.2–10.7) and 207 (160, 302), and in non-severe patients were 2.5 (1.7–3.8) and 149 (110–211), respectively.
Characteristics Patients (N=452) Age (years) Median (IQR) 45.0 (33.0–57.0) Distribution [n (%)] <40 166 (36.7) 40–59 192 (42.5) ≥60 94 (20.8) Sex [n (%)] Male 230 (50.9) Female 222 (49.1) BMI (kg/m2) Median (IQR) 24.3 (21.5–26.4) Distribution [n/N (%)] <18.5 23/368 (6.3) 18.5–23.9 146/368 (39.7) 24–27.9 147/368 (39.9) ≥28 52 /368 (14.1) Wuhan-related exposure [n (%)] Yes 134 (29.6) Close history to COVID-19 cases [n (%)] Yes 285 (63.1) Comorbidities [n (%)] Any 153 (33.8) Hypertension 82 (18.1) Diabetes 37 (8.2) Cardiovascular disease 17 (3.8) Stroke 13 (2.9) Others 36 (23.6) Clinical Classification [n (%)] Mild 41 (9.1) Moderate 339 (75.0) Severe 54 (11.9) Critical 18 (4.0) Signs and symptoms on admission [n (%)] Fever 170 (37.6) Cough 277 (61.3) Dry cough 156 (34.5) Fatigue 170 (37.6) Shortness of breath 63 (13.9) Diarrhea 35 (7.7) Note: Others of comorbidities included pulmonary tuberculosis, chronic bronchitis, emphysema, hepatitis, depression, etc.
Abbreviations: IQR=interquartile range; BMI=body mass index.Table 1. Characteristics of the patients enrolled.
Each one-unit (e.g., from 2 to 3) increase of NLR and each 10-unit increase of PLR was associated with 7% and 1% higher odds of being a severe patient, respectively (adjusted for age, sex, BMI, comorbidity, and onset symptoms, P<0.01). The odds ratios and 95% confidence intervals (OR, 95% CI) for being a severe patient in age groups of <40, 40–59, and ≥60 years were 1.30 (1.13–1.50), 1.04 (1.01–1.08), and 1.09 (0.99–1.20) for NLR, and 1.05 (1.01–1.09), 1.00 (1.00–1.01), and 1.01 (0.97–1.04) for PLR, respectively.
The area under curve (AUC) for predicting severe illness was 0.75 (95% CI: 0.69–0.82) for NLR and 0.67 (0.59–0.74) for PLR in all patients (Figure 1-A). The AUCs in male and female were similar to that in all patients. After sub-analyses by age, the AUC in age groups of <40, 40-59, and ≥60 years were 0.80 (0.64–0.95), 0.75 (0.64–.87), and 0.68 (0.56–0.80) for NLR, respectively, and 0.87 (0.78–0.86), 0.67 (0.56–0.79), and 0.54 (0.42–0.66) for PLR, respectively (Figure 1). The ideal cut-off values for predicting severe COVID-19 infection in patients less than age 40 for NLR and PLR were 3.1 and 192.
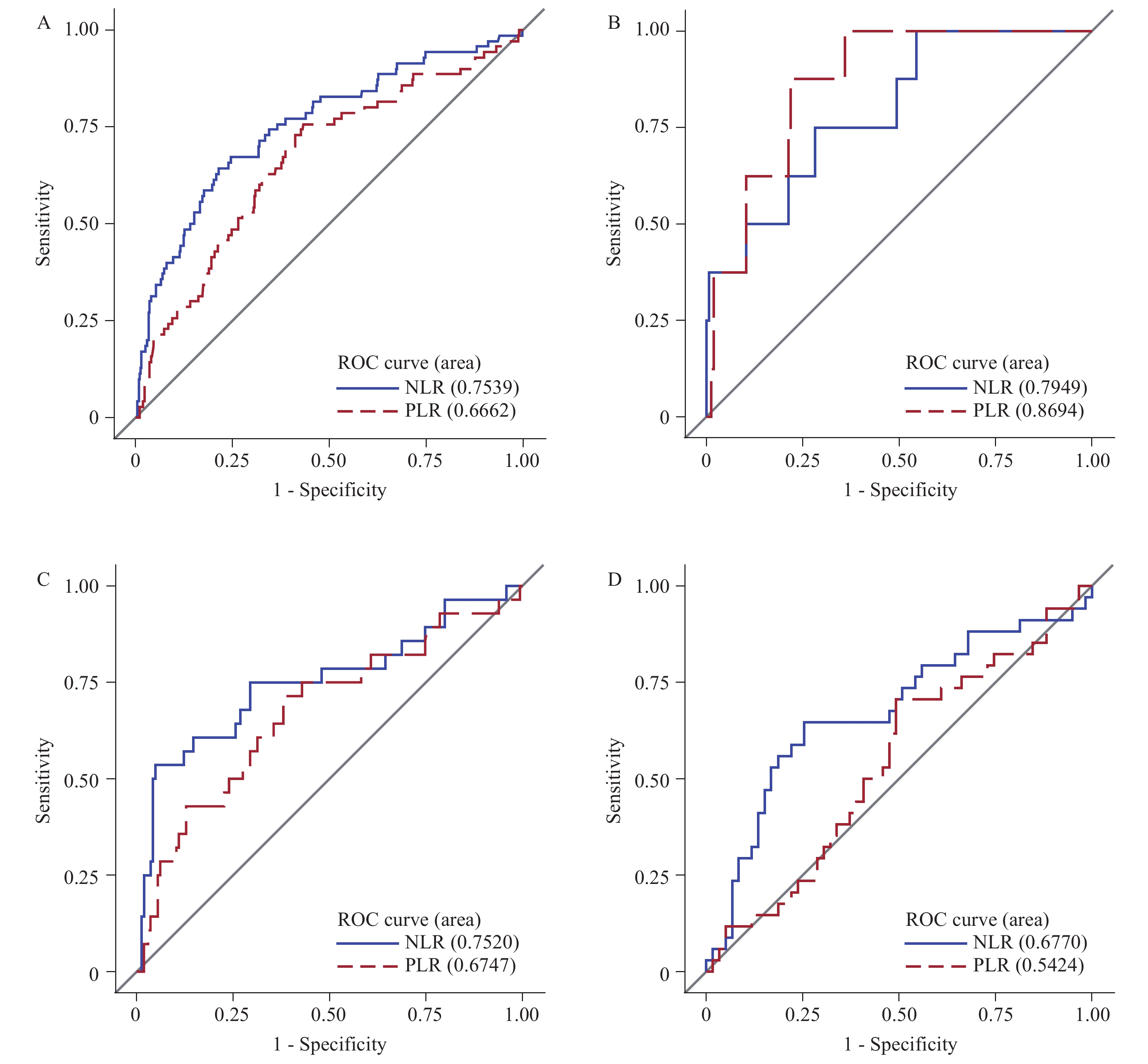 Figure 1.
Figure 1.The area under curve for predicting severe COVID-19 infection for NLR and PLR in (A) all COVID-19 patients, (B) COVID-19 patients <40 years, (C) 40–59 year, and (D) ≥60 years.
Note: abbreviations: ROC=receiver operating characteristic; NLR=neutrophil-lymphocyte ratio; PLR=platelet-lymphocyte ratio. The blue curve represents the ROC of NLR and the red curve represents the ROC of PLR. -
These findings indicate that both NLR and PLR were associated with clinical severity of COVID-19 infection. Higher NLR and PLR were useful predictors in diagnosis and early recognition of severe illness in younger patients of age <40 years. The benefits of using NLR and PLR measurements are because they are simple, rapid, and inexpensive, while also being associated with less patient discomfort, as only peripheral blood samples are required for testing. Furthermore, these values are easily evaluated in most hospital laboratories (8).
This study was subject to some limitations. Because we collected data from medical records, some demographic variables with missing values were not included, such as occupation, education level, and smoking status. This may cause some residual bias. Also, we only used the measurement of NLR and PLR upon admission. Thus, the trajectory of NLR and PLR and their association with clinical course could not be analyzed.
In conclusion, neutrophil, lymphocyte, and platelet counts are a part of routine blood tests, and NLR and PLR values can both be acquired in just five minutes. Because of this, NLR and PLR are recommended as indicators to identify severe COVID-19 patients, especially in young patients under 40 years old. This may help facilitate effective care and prioritize medical resources during a COVID-19 outbreak.
-
No conflicts of interest reported.
-
All participants from 41 hospitals (Supplementary Table S1) in the study.
Number PLADs List of hospitals 1 Shaanxi Shangluo Central Hospital 2 Heilongjiang The People’s Hospital of QiTaiHe 3 Shaanxi Xianyang Central Hospital 4 Anhui The First Affiliated Hospital of Anhui University of Traditional Chinese Medicine 5 Hebei Langfang Hospital of Chinese Medicine 6 Hebei Xingtai Hospital of Chinese Medicine 7 Guangxi The People’s Hospital of GuangXi Zhuang Autonomous Region 8 Guangxi The First People’s Hospital of Fangchenggang 9 Sichuan Mianyang Hospital of Traditional Chinese Medicine 10 Guangxi Liuzhou People’s Hospital 11 Sichuan Affiliated Hospital of North Sichuan Medical College 12 Sichuan The Public Health Clinical Center of Chengdu 13 Hebei Shijiazhuang Fifth Hospital 14 Shanxi The Fourth People’s Hospital of Taiyuan 15 Sichuan The First Hospital of Suihua City 16 Shaanxi Ankang Hospital of Traditional Chinese Medicine 17 Guangxi Beihai Hospital of Chinese Medicine 18 Heilongjiang Harbin Infectious Disease Hospital 19 Hebei Chengde Hospital of Traditional Chinese Medicine 20 Shanxi Datong Fourth Hospital 21 Sichuan Suining Central Hospital 22 Shanxi Jinzhong Infectious Disease Hospital 23 Shanxi Jincheng People’s Hospital, Jincheng 24 Shaanxi Hanzhong Central Hospital, Hanzhong, 25 Shanxi Shuozhou People’s Hospital, Shuozhou 26 Heilongjiang Mudanjiang Kangan Hospital, Mudanjiang 27 Shanxi Xinzhou People’s Hospital, 28 Shanxi Daqing Second Hospital 29 Heilongjiang Jiamusi Infectious Disease Hospital 30 Shaanxi Hanzhong Hospital for Infectious Diseases 31 Shaanxi Shaanxi Infectious Disease Hospital 32 Shaanxi Baoji Central Hospital 33 Shaanxi Xi'an Chest Hospital 34 Heilongjiang Qiqihar Institute for The Prevention and Treatment of Infectious Diseases 35 Shanxi Fenyang Hospital of Shanxi Province 36 Heilongjiang Shuangyashan People’s Hospital 37 Heilongjiang The Greater Khingan Range People’s Hospital 38 Guangxi The Fourth People’s Hospital of Nanning 39 Shanxi The Third People’s Hospital of Linfen 40 Hebei Hengshui Hospital of Chinese Medicine 41 Heilongjiang The First Hospital of Qiqiha Abbreviation: PLADs=provincial-level administrative divisions. Table S1. List of 41 hospitals in the multi-center observational study.
HTML
| Citation: |




 Download:
Download:




