-
Monkeypox (MPX) is an emerging zoonotic disease caused by the MPX virus (MPXV), which is recognized as the most important Orthopoxvirus infection in humans in the smallpox post-eradication era (1). The unexpected increase in human MPX cases in non-endemic countries raises concerns regarding a novel global public health threat (2). Between January 1 and November 10, 2022, a cumulative total of 79,151 laboratory-confirmed cases of MPX and 49 related deaths were reported to the World Health Organization by 110 countries or areas worldwide (3).
The first imported case of MPX in the mainland of China was confirmed on September 16, 2022, in a 29-year-old salesman of Chinese nationality who visited Germany (4). On September 9, the patient had a dry and itchy throat, a fever, and red rashes and pustules displayed on the right thigh. Clinical manifestations were reported, and complete genomes of the MPXV from clinical samples suggest that the MPXV strain in this case belongs to the B.1 branch of the West African lineage (4). The response to this public health emergency must include rapid isolation and identification of MPXV and development of novel assays for MPXV (5). To date, MPXV isolation and characterization have not been reported in the mainland of China. Additionally, limited information is available regarding a plaque assay for an infectious titration of MPXV and a plaque reduction neutralization test (PRNT) for MPXV-neutralizing antibody (nAb) detection on the basis of the MPXV-B.1 strains of the current epidemic.
In this study, the isolation and characteristics of MPXV in the first imported case of MPX in the mainland of China are reported. A plaque assay for MPXV infectious titration and a PRNT for MPXV nAb detection were developed.
-
For virus isolation, clinical specimens including skin blister fluid, oropharyngeal and nasopharyngeal swabs, and blood were collected 6 days after symptom onset and sent to the National Institute for Viral Disease Control and Prevention, China CDC. MPXV isolation was performed in biosafety level 3 laboratories on Vero cells. The specimens were diluted at a 1∶5 ratio with culture medium, which contained 2% fetal bovine serum-Modified Eagle’s Medium (FBS-MEM) and 1% antibiotics (penicillin 5,000 IU/mL, streptomycin 2,500 μg/mL, and amphotericin B 10 μg/mL, brief as PSA), treated samples were kept at 25 °C for 30 min and inoculated onto Vero cells. After incubation for 6 h at 37 °C in 5% CO2, the supernatant was replaced with fresh medium. Cytopathic effect (CPE) appearance was observed daily.
A quantitative real-time polymerase chain reaction (qPCR) assay was conducted to confirm virus replication. Cell supernatants or lysates were collected and qPCR of MPXV was performed as reported previously (6).
For an indirect immunofluorescence assay, Vero cells were grown overnight in 8-well plates until 80% confluence and infected with the first passage of MPXV isolate. The cells were fixed with 4% paraformaldehyde 48 h post-infection, rinsed twice with phosphate-buffered saline (PBS), permeabilized with PBS containing 0.05% Triton X-100 (PBST) for 10 min, and blocked in PBST containing 2% goat serum for 30 min. After three washes, the cells were incubated with serum from the MPX case and diluted to 1∶100 for 1 h at 37 °C and then incubated with the FITC-labelled anti-human immunoglobulin G diluted to 1∶200, stained with 4’6-diamidino-2-phenylindole (DAPI) for 5 min, and were finally examined using the Olympus IX73 inverted fluorescence microscope.
MPXV morphologic features were determined by transmission electron microscopy assays. For the negative staining of virus particles in supernatant, the samples were enriched by ultracentrifugation and absorbed on film-coated grids, following staining with 1% PTA. For the ultrathin section sample, cell pellets were collected 48 h post infection, fixed with 2.5% paraformaldehyde-2% glutaraldehyde in 0.1 mol/L cacodylate buffer (pH 7.4) and 1% osmium tetroxide, respectively, following dehydration, infiltration with epoxy resin and polymerization. Ultrathin sections (60–80 nm) were cut with an ultramicrotome from the resin block embedded with samples and stained with uranyl acetate and lead citrate. Finally, the grids and stained sections were observed and photographs were taken under a Tecnai 12 transmission electron microscope (FEI Co., USA) at 120 kV.
Plaque assays were developed to quantify the infectious virus titer. The Vero cells were seeded on 12-well plates overnight. Ten-fold serial dilutions were performed in MEM medium containing 2% FBS and 1% antibiotics of PSA. 400 µL was used per well to infect a monolayer of Vero cells. 72 to 96 h later, the cells were fixed with 4% paraformaldehyde and stained with crystal violet. Plaque forming units (PFUs) per mL were calculated on the basis of the number of plaques multiplied by the dilution factor.
To identify the nAb induced after MPXV infection, a PRNT assay based on MPXV-B.1-China-C-Tan-CQ01 was developed. The Vero cells were seeded in 12-well. Sera were collected from the patient with MPX on day 6 after symptom onset and from two healthy 30-year-old individuals as controls, inactivated for 30 min at 56 °C, diluted in 1∶20 at the beginning, and subsequently serially two folds diluted. MPXV stock (100 PFU) was added. The virus-serum mix was then incubated at 37 °C for 1 h and added to the Vero cells. The Vero cells were incubated for 96 h, fixed with 4% paraformaldehyde and stained with crystal violet.
-
Swabs from the skin lesion, oropharynx, nasopharynx, and whole blood samples were used for viral isolation in the Vero cells. Viral replication was confirmed by qPCR assay based on conserved F3L genes that specifically detect MPXV with DNA from cell cultures collected at 48, 96, 120, and 144 h after inoculation (Table 1). The cycle threshold (Ct) values for the nasopharyngeal swabs and blood incubations were negative at 48 h and 144 h. In contrast, for the blister fluid swab inoculation, Ct values of 27.88, 19.05, and 18.15 were detected at 48 h, 96 h, and 120 h after the inoculation. For the oropharyngeal swab inoculation, Ct values were detected as 31.00, 27.28, and 25.68 at 48 h, 96 h, and 144 h, respectively. These results indicated that MPXV replicated in the blister fluid and oropharyngeal incubation.
Specimen Ct values* CPEs at harvest time MPXV isolation Clinical sample 48 h 96 h 120 h 144 h Blister fluid swab 20.76 27.88 19.05 18.15† ND ++++ Positive Oropharyngeal swab 27.81 31.00 27.28 ND 25.68† + Positive Nasopharyngeal swab 31.00 ND ND ND ND† - Negative Blood 33.65 ND ND ND ND† - Negative Note: CPEs: -, Negative or no CPEs; +, 0–25%; ++, 25%–50%; +++, 50%–75%; ++++, 75%–100%.
Abbreviation: MPXV=monkeypox virus; qPCR=quantitative real-time polymerase chain reaction; ND=not determined; CPEs=cytopathic effects; Ct=cycle threshold.
* For the blister fluid swab, viral DNAs from the supernatant of 48 h and 96 h were detected, while DNA from cell lysates of 120 h was detected. For the oropharyngeal swab, nasopharyngeal swab and blood samples, viral DNAs from the supernatant of 48 h, 96 h, 120 h were detected, while DNA from cell lysates of 144 h was detected.
† Harvest cell lysate.Table 1. Detection of monkeypox virus (MPXV) DNA using quantitative real-time polymerase chain reaction in Vero cell cultures with different specimens from the MPX case.
The virus-induced changes in CPE-related cell morphology were observed daily. The nasopharyngeal and blood-incubated cells were CPE-negative. In contrast, CPEs were observed within 48 h for the skin blister fluid-incubated cells, exhibiting cell rounding, detachment, or even death (Figure 1A). The degree of visible damage to the cells increased at 72 h, 96 h, 120 h, and 144 h, resulting in >85% CPEs (Table 1). Meanwhile, minor CPEs were also observed after 48 h for the oropharyngeal incubated cells, resulting in <20% CPEs at 144 h post-incubation (Table 1).
 Figure 1.
Figure 1.Characterization of MPXV-B.1-China-C-Tan-CQ01 in the Vero cell. (A) Cytopathic effects of monkeypox virus (MPXV) in the Vero cell; (B) Immunofluorescence analysis of Vero cell infected or uninfected with MPXV-B.1-China-C-Tan-CQ01 using the serum of the MPX case; (C) Transmission electron microscopy micrographs of MPXV.
Note: In panel A, the typical cytopathic effects are observed in the Vero cells 48 h after the first passage, thereby exhibiting cell detaching and rounding. In panel B, the nuclei were stained with DAPI (blue), while the MPXV infected cell showed green fluorescence. In panel C, the left panel showed the negative staining of purified MPXV particles with a typical brick shape observed in a diameter that varies between 200 nm and 300 nm and irregular tubular strips on the surface. The right panel showed the ultrathin section of MPXV infected Vero cell and mature virus particles in cytoplasm that show typical morphology including a dumbbell shaped core and the outer membrane on longitudinal orientation.
Abbreviation: MPXV=monkeypox virus; MPX=monkeypox; DAPI=4'6-diamidino-2-phenylindole.
Immunofluorescence assay with the patient’s serum was performed to confirm MPXV antigen expression, which showed the infected foci, visualized with green fluorescence in the cell, reacted with the serum of the patient with MPX (Figure 1B). IFA with anti-rabbit antibody against orthopoxviruses was also positive around the infected foci (data not shown).
Negative stained virus particles present a typical brick shape about 200 nm × 200 nm × 250 nm in size with irregular tubular strips on the surface. On sections, mature virus particles in cytoplasm show typical morphology including a dumbbell-shaped core and the outer membrane on longitudinal orientation (Figure 1C).
Plaque assays indicated that the infectious MPXV titers in the first passage of blister fluid or oropharyngeal swab incubation were 6 × 104 PFU/mL and 10 PFU/mL, increased to 1 × 105 PFU/mL and 1 × 104 PFU/mL for the second passage, and to >106 PFU/mL after the third passage (Figure 2A).
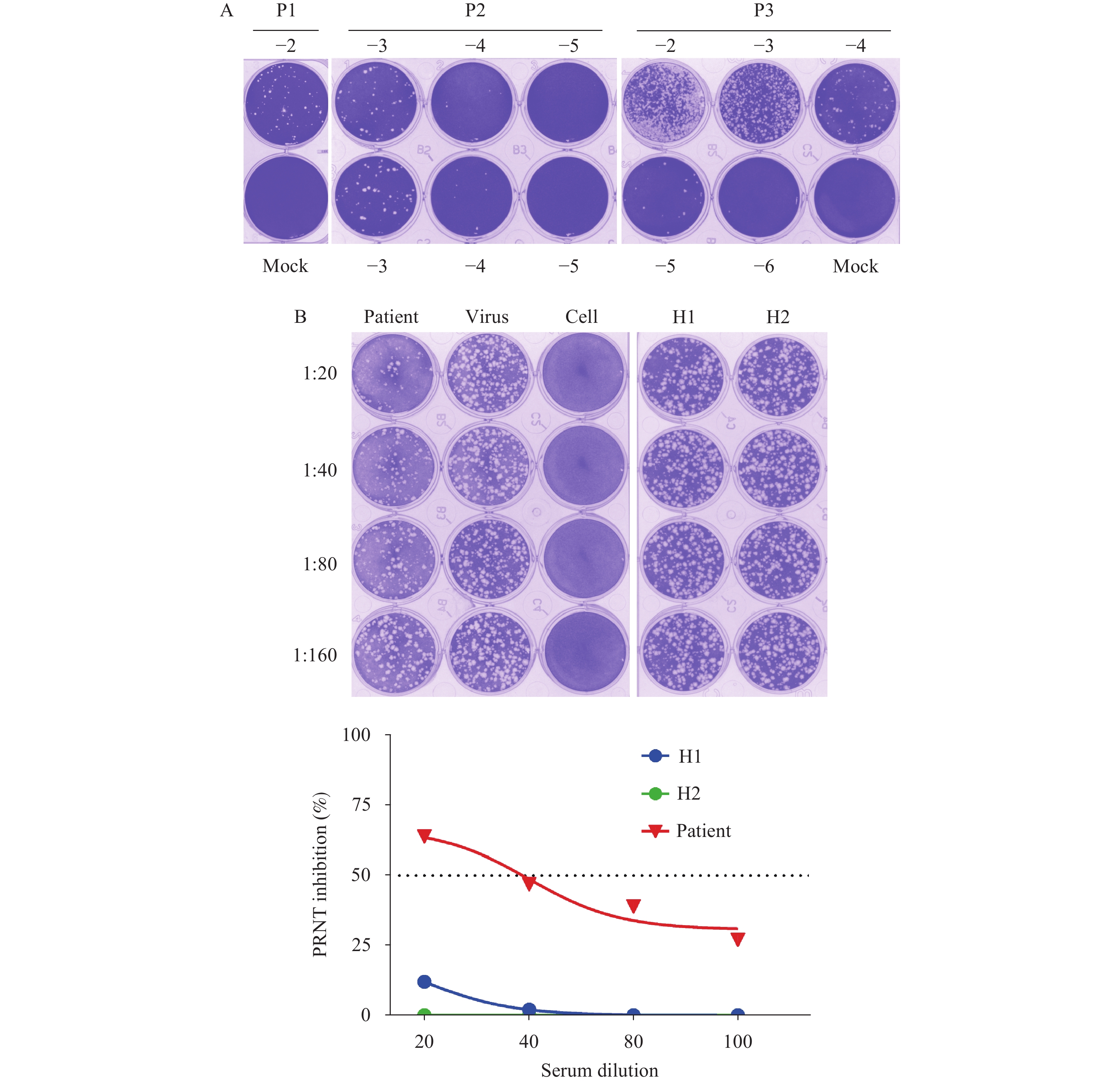 Figure 2.
Figure 2.Plaque assays and PRNT based on MPXV-B.1-China-C-Tan-CQ01. (A) Quantification of infectious monkeypox virus from clinical samples (the first passage of isolation, P1, to the third passage of isolation, P3, using plaque assay). (B) Plaque reduction neutralization test to detect neutralizing antibodies (nAb) against MPXV in the serum of the MPX case (patient) and two 30-year-old healthy donors (H1 and H2).
Note: In panel A, the plates are fixed and stained with crystal violet 72 h to 96 h post-infection. Representative plates for the MPXV titration from blister fluid are shown. Plaques for P1 with a dilution of 10−2 (−2) and mock, for P2 with a dilution from 10−3 to 10−5 (−3, −4, −5) in double well, and for P3 with a dilution from 10−2 to 10−6 (−2, −3, −4, −5, −6) and mock are all shown. In panel B, the representative plate and neutralization cures are presented.
Abbreviation: MPXV=monkeypox virus; MPX=monkeypox; P1=the first passage of isolation; P2=the second passage of isolation; P3=the third passage of isolation; PRNT=The plaque reduction neutralization test; nAb=the neutralizing antibody; H1=healthy donor 1; H2=healthy donor 2.
A PRNT in the Vero cells with the MPXV-B.1-China-C-Tan-CQ01 was established. Serum 50% neutralization titer (NT50) was detected as 35 for the MPXV case 6 days after symptom onset, while negative for the 30-year-old healthy donors (Figure 2B).
-
In this study, the isolation and characteristics of MPXV from the first imported MPX case in the mainland of China in 2022, named MPXV-B.1-China-C-Tan-CQ01, were reported. Based on a review of current literature, this is the first reported MPXV isolate in the mainland of China. Additionally, MPXV replication using qPCR, CPEs, and the morphological features in the Vero cells were characterized. Furthermore, novel plaque assays and PRNT were developed with the MPXV-B.1-China-C-Tan-CQ01 to quantify the infectious MPXV and nAb titer in the MPX case.
Replication-competent MPXV isolated from skin lesion swabs, oropharyngeal swabs, anal swabs, urethral swabs, and semen has been reported previously (5,7-11). However, MPXV isolation using oropharyngeal or nasopharyngeal swabs was rarely reported (12-13). Interestingly, even in the absence of oral lesions in the patient, replication-competent MPXV was also isolated from the oropharyngeal swabs in the study. This is indicative of a sufficient infectious virus presenting in the oropharyngeal site, leading to possible passage by oral droplets and contact. The results support the notion of the high prevalence of oropharyngeal and perioral lesions in the current MPX outbreak.
Current knowledge is limited to MPXV shedding and the correlation between the detected Ct value and infectious MPXV load in clinical specimens. A previous report indicated that MPXV DNA levels correlated with infectivity in clinical samples and defined a threshold (Ct ≥35; viral DNA ≤4,300 copies/mL) that predicts poorly infectious specimens (14). The study measured the infectious potential of the first passages of virus isolation, with a Ct value of 20.76 for the skin blister fluid swab, the infectious MPXV was 6 × 104 PFU/mL. With a Ct value of 27.81 for the oropharyngeal swabs, the infectious MPXV was 10 PFU/mL. A clear correlation between the Ct value and virus isolation needs to be investigated in further studies, with more samples collected to represent different disease course times and body fluids.
Neutralization assays are often used to detect nAbs and determine a possible protective antibody titer after infection or vaccination. To date, data on nAbs in vaccinated individuals or MPXV-infected cases against MPX are rare. Herein, the study described a PRNT assay based on MPXV-B.1-China-C-Tan-CQ01 and detected MPXV-nAbs in the patient’s serum collected on day 6 after symptom onset. As the patient was 29 years old, there was no smallpox vaccination history, and the results indicated that MPXV-infected individuals could rapidly induce nAb (<2 weeks). The PRNT described in the study is able to monitor the neutralizing immune response to MPXV and determine whether the nAb induced by historic smallpox vaccination is cross-reactive with MPXV.
In summary, the first MPXV strain in the mainland of China, named MPXV-B.1-China-C-Tan-CQ01 and representative of the current epidemic MPXV B.1 clade, was isolated. Characterization of the CPEs, replication, and morphologic features of MPXV B.1 improves collective understanding of the current MPX outbreak and its control. The novel plaque assay and PRNT developed in this study will pave the way for further research on MPXV as well as the development of anti-viral drugs and vaccines against the MPX epidemic.
-
No conflicts of interest.
HTML
| Citation: |




 Download:
Download:




